The diagram shows some allotropes of carbon. a. Give the name of allotrope a, which has the
Question:
The diagram shows some allotropes of carbon.
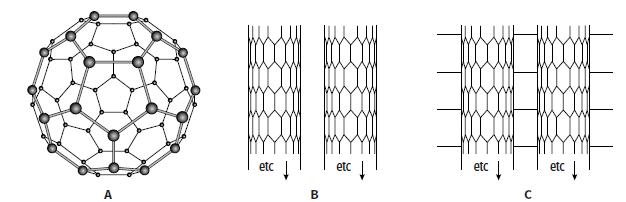
a. Give the name of allotrope a, which has the formula C60.
b. Explain in terms of structure and bonding why structure a is gaseous at 800 °C but diamond is not.
c. Structure B shows an allotrope of carbon in the form of tubes.
i. Give the name of this allotrope of carbon.
ii. Describe the similarities and differences between structure B and graphite.
d. Structure c is stronger than structure B when a force is applied in the same direction as the long axis of the tube. Explain why structure C is stronger.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:





