se the cell voltage method described in Worked examples 6 and 7 above to answer question 21,
Question:
se the cell voltage method described in Worked examples 6 and 7 above to answer question 21, parts a to d.
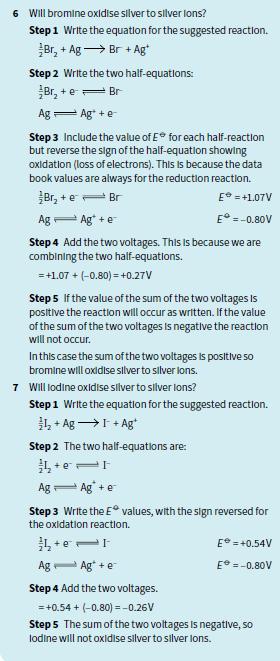
Data From Question 21:
Use the data in Appendix 2 (page 474) to predict whether or not the following reactions are feasible. If a reaction does occur, write a balanced equation for it.
a Can MnO4 – ions oxidise Cl– ions to Cl2 in acidic conditions?
b Can MnO4– ions oxidise F– ions to F2 in acidic conditions?
c Can H+ ions oxidise V2+ ions to V3+ ions?
d Can H+ ions oxidise Fe2+ ions to Fe3+ ions?
Data From Appendix 2:

6 Will bromine oxldise silver to silver lons? Step 1 Write the equation for the suggested reaction. Br, + Ag → Br + Ag Step 2 Write the two half-equations: Br, + e= Br Ag 一Ag +e Step 3 Include the value of E* for each half-reaction but reverse the sign of the half-equation showing oxidation (loss of electrons). This Is because the data book values are always for the reduction reaction. Br, + e- Br E = +1.07V Ag Ag* +e E° =-0.80V Step 4 Add the two voltages. This Is because we are combining the two half-equations. =+1.07 + (-0.80) = +0.27V Step 5 lf the value of the sum of the two voltages Is positive the reaction will occur as written. If the value of the sum of the two voltages is negative the reaction will not occur. Inthis case the sum of the two voltages is positive so bromine will oxidise slver to slver lons. 7 Will lodine oxidise silver to silver lons? Step1 Write the equation for the suggested reaction. 弘+ Ag→I+Ag Step 2 The two half-equations are: + e Ag- Ag +e Step 3 Write the E values, with the sign reversed for the oxidation reaction. E = +0.54V Ag- Ag* +e E =-0.80V Step 4 Add the two voltages. = +0.54 + (-0.80) =-0.26V Step 5 The sum of the two voltages Is negative, so lodine will not oxidise silver to silver lons.
Step by Step Answer:
To determine whether the given reactions are feasible we can use the cell voltage method This involv...View the full answer



Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Sciences questions
-
Examples 2 and 6 described a study about therapeutic touch (TT). A second run of the same experiment in the study used 13 TT practitioners who had to predict the correct hand in each of 10 trials. a....
-
A silver oxidezinc cell maintains a fairly constant voltage during discharge (1.60 V). The button form of this cell is used in watches, hearing aids, and other electronic devices. The half-reactions...
-
In Exercises 7 and 8 of Chapter 4 you worked with data on sales for a line of skiwear that is produced by HeathCo Industries. Barbara Lynch, product manager for the skiwear, has the responsibility of...
-
Using the information from 2-16, create a cash flow statement for 2013. In 2-16 Bristle Brush-Off Corporation: Income Statements for Years Ended December 31 ($000s) 2013 2012 $7,950 5,100 350 750...
-
Let z be a random variable with a standard normal distribution. Find the indicated probability and shade the corresponding area under the standard normal curve. P( 1.78 z 1.23)
-
A company has a factory that is designed so that it is most efficient (average unit cost is minimized) when producing 15,000 units of output each month. However, it has an absolute maximum output...
-
A magnet lies on a table. You place a second magnet near the first one so that the two repel each other. Identify all the forces exerted on the first magnet.
-
Turner, Roth, and Lowe are partners who share income and loss in a 1:4:5 ratio. After lengthy disagreements among the partners and several unprofitable periods, the partners decide to liquidate the...
-
11.A simple LR circuit is connected to a battery at t = 0. The time instant at which rate of energy storage in inductor is half of power delivered by battery 2L (1) In 2 (3) In 2 (2) In (4) (4) In 3
-
A physics student has a 25% chance of passing their physics final. If 124 randomly selected physics students are selected, answer the following. a) Which is the correct wording for the random...
-
Suggest a suitable reagent that can carry out each of the following oxidations or reductions. Use the data in Appendix 2 to help you. a. The reduction of Zn 2+ ions to Zn. b. The oxidation of Br ...
-
In each of the chemical reactions a to c: i. Which species gains electrons? ii. Which species loses electrons? iii. Which species is the oxidising agent? iv. Which species is the reducing agent? a....
-
We have given the number of possible values for two variables of a population. For each exercise, determine the maximum number of expected frequencies that can be less than 5 in order that Assumption...
-
How can distributive leadership models, characterized by shared decision-making and collaborative problem-solving among team members, enhance organizational adaptability and innovation in...
-
How long will it take $1880.00 to accumulate to $2207.00 at 9% p.a. compounded monthly? State your answer in years and months (from 0 to 11 months)?
-
A dermatologist trainee investigates a certain type of skin cancer by inducing this type of cancer in 25 rats and then treating them with a new type of drug. The number of hours is counted until the...
-
Kia's employer paid for her education during the tax year. the cost of her tuition was a $9500 what is the maximum amount of employer-provided educational assistance that a taxpayer can exclude from...
-
What is the Porter's five forces of Commercial Bank of Qatar with Explanation for each point? What is the vision, mission and goals for Commercial Bank of Qatar with explain each of them? What is the...
-
Consider the emission spectrum of singly ionized helium (He+). Find the longest three wavelengths for the series in which the electron makes a transition from a higher excited state to the first...
-
A number of years ago the United Food and Commercial Workers Union organized 800 workers of the 1035 employees at one of the Wilson Brothers food operations in Toronto, Ontario. The employees include...
-
A sample of Na2SO4(s) is dissolved in 225 g of water at 298 K such that the solution is 0.325 molar in Na 2 SO 4 . A temperature rise of 0.146C is observed. The calorimeter constant is 330. J K 1 ....
-
Assign a name for each of the following compounds. a. b. c.
-
a. Using the relationships derived in Example Problem 7.1 and the values of the critical constants for water from Table 7.2, calculate values for the van der Waals parameters a, b, and R from z c , T...
-
This program doesn't terminate when it's run: #include "csapp.h" int main() { int fdsA[2]; int fds8[2]; char buffer[2]: pid t pid; Pipe(fdsA); Pipe (fdsB); pid = Fork(); if (pid == 0) { Dup2(fdsA[0],...
-
(30 Pts) Write a class called Student. The class must have two fields: name of the String type and gpa of the double type. Write all the necessary constructors, getters, setters, and toString()...
-
Salient features of divide-and-conquer algorithms, derivation of recurrence relation for divide-and-conquer algorithm and the solution of recurrence relation using two approaches, (i) Master theorem...

Study smarter with the SolutionInn App


