Use the data from Table 21.2 to work out the pH values of the following solutions: a.
Question:
Use the data from Table 21.2 to work out the pH values of the following solutions:
a. 0.0200 mol dm–3 aqueous benzoic acid
b. 0.0100 mol dm–3 hydrated aluminium ions
c. 0.100 mol dm–3 aqueous methanoic acid.
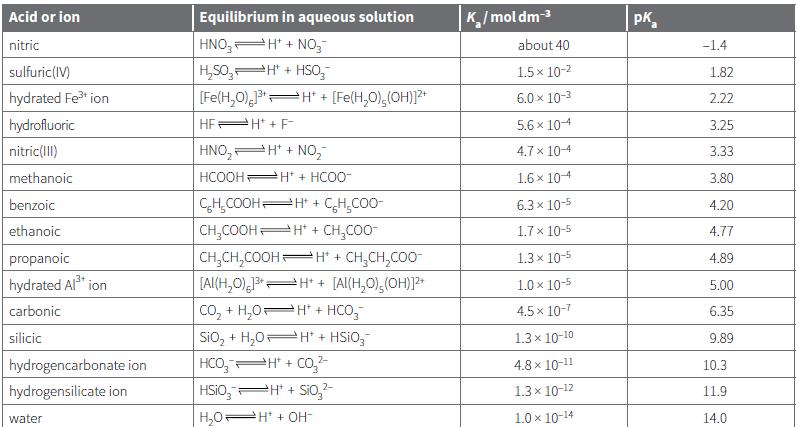
Transcribed Image Text:
Acid or ion Equilibrium in aqueous solution K/moldm-3 pK, nitric HNO, H' + NO,- about 40 -1.4 H,S0, H" + HSo, [Fe(H,0), H" + sulfuric(IV) 1.5 x 10-2 1.82 hydrated Fe ion [Fe(H,O),(OH))?* 6.0 x 10-3 2.22 hydrofluoric HEPH + F- 5.6 x 104 3.25 nitric(1II) HNO, H* + NO, 4.7 x 104 3.33 methanoic HCOOH Ht + HCO0- 1.6 x 104 3.80 CH,COOHH* + CH,CO0- CH,COOH H* + CH,CO0- benzoic 6.3 x 10-5 4.20 ethanoic 1.7 x 10-5 4.77 propanoic CH,CH,COOH H* + CH,CH,COO- 1.3 x 10-5 4.89 [Al(H,0) H* + [Al(H,0),(OH)]? Co, + H,0H* + HCO,- Sio, + H,0 H* + HSIO,- HCO, H + Co,2- hydrated Al* ion 1.0 x 10-5 5.00 carbonic 4.5 x 10-7 6.35 silicic 1.3 x 10-10 9.89 hydrogencarbonate ion 4.8 x 10-11 10.3 hydrogensilicate ion HSIO,PH + Sio,- 1.3x 10-12 11.9 water H,0H* + OH- 1.0 x 10-14 14.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
a The concentration of aqueous benzoic acid is 002 moldm3 ka Value of acid is 631...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
Use the data from The Wall Street Journal in Figure to verify the trin ratio for the NYSE. Is the trin ratio bullish orbearish? Trading Diary: Volume, Advancers, Decliners Markets Diary Issues...
-
Use the data from Exercise 15.
-
Use the data from The Financial Post in Figure 10.8 to construct the trin ratio for the TSX. Is the trin ratio bullish or bearish? Figure 10.8 Market breadth. Market breadth TRADING (1000s) ASWANCE...
-
Energy of the emitted photon when an L-electron drops into the k-state in copper (z = 29) is -1 use R=109737 cm, cm =1.23910eV] 7994.6 eV 1094.6 eV 1293.6 eV 1097.3 eV
-
Sketch the areas under the standard normal curve over the indicated intervals and find the specified areas. To the left of z = 0.47
-
A local fast-food restaurant wants to analyze its drive-thru window. At this time, the only information known is the average number of customers in the system (4.00) and the average time a customer...
-
A 1000-kg compact car and a \(2000-\mathrm{kg}\) van, each traveling at \(25 \mathrm{~m} / \mathrm{s}\), collide head-on and remain locked together after the collision, which lasts 0.20 s. (a)...
-
It certainly is nice to see that small variance on the income statement after all the trouble weve had lately in controlling manufacturing costs, said Linda White, vice president of Molina Company....
-
7. A vertical spring is fixed to one of its end and a massless plank fitted to the other end. A block is released from a height has shown. Spring is in relaxed position. Then choose the correct...
-
Cassi (SSN 412-34-5670) has a cash-basis home cleaning business she runs as a sole proprietorship. The following are the results from business operations for the tax year 2018: Gross receipts $...
-
A buffer solution consists of 6.00 g of ethanoic acid (CH 3 COOH) and 12.3 g of sodium ethanoate (CH 3 COONa) in 200 cm 3 of aqueous solution. (A r values: H = 1.0, C = 12.0, O = 16.0, Na = 23.0; Ka...
-
A saturated solution of copper(I) sulfide, Cu 2 S, contains 1.91 10 12 g of Cu 2 S dissolved in 1 dm 3 of water. (A r values: Cu = 63.5, S = 32.1) a. Write an equilibrium expression for the...
-
Describe how sodium dihydrogen phosphate can be used as a pH buffering agent.
-
5. Consider the inclined manometer below. Plot the change in differential pressure versus the change in differential length, &, of the mercury in the inclined section. SG=0.9 30 100 mm 50 mm Water 80...
-
Considering the principles of interpersonal communication, which of the principles is the most important to you and why? Which of the principles of interpersonal communication do you struggle with...
-
A common trick for a show dog is to jump vertically upward off its hind legs. A particular dog of mass = 1 1 . 6 k g m=11.6 kg shows off its jumping ability by jumping straight up and rising = 0 . 4...
-
You estimate that the dividend of stock AB will be $1.05/ share at YE 2024, then continue to grow in perpetuity at 3% per year. If AB's discount rate is 8%,what do you estimate the present value of...
-
What steps can you take to ensure your early childhood environment is inclusive allowing for access to all children? What information presented this week will help you to design an inclusive...
-
What is the maximum wavelength of a photon that can create an electron-positron pair?
-
Chris Zulliger was a chef at the Plaza Restaurant in the Snowbird Ski Resort in Utah. The restaurant is located at the base of a mountain. As a chef for the Plaza, Zulliger was instructed by his...
-
Identify the reactants that you would use to make each of the following enamines: (a) (b) (c) -N
-
Predict the product of the two-step procedure below, and draw a mechanism for its formation: 1) [H1, -N-NH2 -H2 2) / -0, heat
-
Propose a plausible mechanism for each of the following hydrolysis reactions: (a) (b) (c) (d) EtO OEt * * + 2 ELOH (b) N. * .N'
-
The sticker price as displayed on a new automobile is $35,450. After a lengthy negotiation, you bought the car for $32685. Determine the percentage reduction off the sticker price that you are...
-
Divide. 2 x +7xy+6y 2x-18y x+4xy-12y 1 x-9y Simplify your answer as much as possible.
-
The price of oil recently went from $13.00 to $18.20 per case of 12 quarts. Find the ratio of the increase in price to the original price. The ratio is (Type a simplified fraction.)

Study smarter with the SolutionInn App


