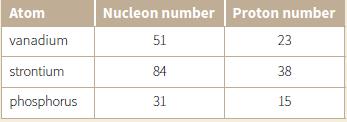
Use the information in the table to deduce the number of electrons and neutrons in a neutral
Question:
Use the information in the table to deduce the number of electrons and neutrons in a neutral atom of:
a. Vanadium
b. Strontium
c. Phosphorus.
Transcribed Image Text:
Atom Nucleon number Proton number vanadium 51 23 strontium 84 38 phosphorus 31 15
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
element Nucleon Number or Mass number ...View the full answer

Answered By

Konda Venkatesham
I'm having 32+ years of teaching experience in dealing chemistry for high school, college levels and entrance exams like EAMCET, EE, and NEET.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
In each part, use the information in the table to determine whether the linear system Ax = b is consistent. If so, state the number of parameters in its general solution. 3x3 3x3 3x3 5x9 5x9 4x4 6x2...
-
Use the information in Table 2.5 to predict the standard reaction enthalpy of2 H2 (g) + 02(g) 2 H2O (1) at 100C from its value at 25C.
-
Use the information in Table 18.2 on page 602 to calculate the total federal income tax paid, the marginal tax rate, and the average tax rate for people with the following incomes. (For simplicity,...
-
Express the following sum in closed form. 2 k 2 (4 + 3 - 4 ) = n k 1
-
Determine the mass of each substance. a. Na b. B2O3 c. S2Cl2
-
Terry Manufacturing Company was started on January 1, 2015, when it acquired $2,500 cash by issuing common stock. During its first year of operation, it purchased $500 of direct raw materials with...
-
The annual sales for Saudi Aramco Corporation were $4.7 million last year. All sales are on credit. The firms end-of-year balance sheet was as follows: The firms income statement for the year was as...
-
Mortar Corporation acquired 80 percent ownership of Granite Company on January 1, 20X7, for $173,000. At that date, the fair value of the noncontrolling interest was $43,250. The trial balances for...
-
why is there an OSI and TCP / IP model?
-
In a two-dimensional cylindrical configuration the radial (r) and angular () spacing's of the nodes are uniform. The boundary at r = ri is of uniform temperature Ti. The boundaries in the radial...
-
Zirconium, Zr, and hafnium, Hf, are metals. An isotope of zirconium has 40 protons and 91 nucleons. a. i. Write the isotopic symbol for this isotope of zirconium. ii. How many neutrons are present in...
-
A beam of electrons is passing close to a highly negatively charged plate. When the electrons pass close to the plate, they are deflected (bent) away from the plate. a. What deflection would you...
-
A steel pipe of \(100 \mathrm{~mm}\) bore and \(10 \mathrm{~mm}\) wall thickness, carrying steam at \(250^{\circ} \mathrm{C}\), is insulated with \(30 \mathrm{~mm}\) of a molded high-temperature...
-
Fewer workers are in traditional, full-time stable jobs while a growing number are in so-called contingent jobs, such as temporary or on-call positions, or are treated as independent...
-
Earnhardt Driving School's 2018 balance sheet showed net fixed assets of $5.4 million, and the 2019 balance sheet showed net fixed assets of $6 million. The company's 2019 income statement showed a...
-
Money is deposited into a fund continuously for 2n years with the amount per year being 2000. The accumulated amount at the end of 2n years is 68934.44616, and the accumulated amount at the end of n...
-
Under fixed-interval lot sizing, order sizes for component parts are determined based on which one of the following? Gross requirements - net requirements for a predetermined number of periods Net...
-
Should technical violations of probation and parole be used as a cause to re-incarcerate someone? Please justify your position and defend your answers with facts/data/research.
-
When 3-methyl-1-butene reacts with HBr, two alkyl halides are formed: 2-bromo-3 methylbutane and 2-bromo-2-methylbutane. Propose a mechanism that explains the formation of these products.
-
Sheldon and Leonard had a million-dollar idea. In order to make it happen, they have to do special research first. Only Kripke can help them in this matter. But Kripke is known to be the first-class...
-
The barrier to rotation of bromoethane is 15 kJ/mol. Based on this information, determine the energy cost associated with the eclipsing interaction between a bromine atom and a hydrogen atom.
-
Menthol, isolated from various mint oils, is used in the treatment of minor throat irritation. Draw both chair conformations of menthol, and indicate which conformation is lower in energy. Menthol
-
Draw both chair conformations for each of the following compounds. In each case, identify the more stable chair conformation: (a) Methylcyclohexane (b) Trans-1,2-Diisopropylcyclohexane (c)...
-
Explain when in business it "makes sense" to recommend Long-Term Debt vs Lease Financing when developing/expanding a product?
-
We discussed and learned about the 4P's this week. Consider a time when you got to be mindful of a modern item or benefit, and the circumstances around it. Clarify how innovation impacted how you...
-
Pollen describes American farmers today as "the most productive humans who have ever lived". Explain what he means by this. Discuss why most of these farmers can't earn enough or eat enough off their...

Study smarter with the SolutionInn App


