a. Show that the intrinsic stability analysis for fluid equilibrium at constant temperature and volume leads to
Question:
a. Show that the intrinsic stability analysis for fluid equilibrium at constant temperature and volume leads to the single condition that
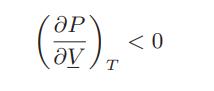
b. Show that intrinsic stability analysis for fluid equilibrium at constant temperature and pressure does not lead to any restrictions on the equation of state.
Transcribed Image Text:
ӘР ( ) .. ᎧᏙ <0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Intrinsic Stability Analysis for Fluid Equilibrium at Constant Temperature and Volume Intrinsic stability refers to the stability of a system with r...View the full answer

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
In some cases if pure liquid A and pure liquid B are mixed at constant temperature and pressure, two liquid phases are formed at equilibrium, one rich in species A and the other in species B. We have...
-
A student is using a model to show how charge is induced. A balloon that was rubbed on animal fur and charged negatively is brought near two aluminum cans resting on two separate cups. Draw (-) signs...
-
A box (m) of mass 60 kg is pulled across a rough floor with a force (F) of 299 N.. The coefficient of friction is 0.23. Determine the acceleration. Record to the nearest 0.01 m/s. Use g = 10 m/s
-
Design a dam structure (your choice of shape and size) that will collect water and will be used for water supply, power generation, and flood control. Consider that the maximum water surface level...
-
Acme Realty, a real estate development company, is a limited partnership organized in Georgia. It is planning to develop a two hundred- acre parcel of land for a regional shopping center and needs to...
-
The equilibrium constant for the reaction \(\mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g}) ightarrow 2 \mathrm{NH}_{3}\) is 0.1084 . Under the same conditions, the equilibrium constant for...
-
For a system with periodic motion, the trajectory in the phase plane is a(n) a. closed curve b. open curve c. point
-
Selected comparative financial statements of Haroun Company follow. Required 1. Compute trend percents for all components of both statements using 2008 as the base year. (Round percents to one...
-
PCU Corporation's checkbook balance on December 31, 2015 was P160,000. On the same date, PCU held the following items in its safe: A P50,000 check payable to PCU, dated January 2, 2016, was not...
-
Shah Ltd (Shah) manufactures a single product, the Choco. The budgeted sales price and production cost per unit of the Choco is as follows: Selling price 360 Variable materials 50 Variable labour 40...
-
a. Show that the conditions for vapor-liquid equilibrium at constant N , T, and V are G V = G L and P V = P L . b. Show that the condition for vapor-liquid equilibrium at constant N , T, and P is G V...
-
a. Show that the condition for equilibrium in a closed system at constant entropy and volume is that the internal energy U achieve a minimum value subject to the constraints. b. Show that the...
-
You want to save money from your business operation to replace a truck that has been used in delivery. The truck will be replaced after 10 years from now and the replacement cost would be about...
-
Gleed argues he had two disabilities for which he needed accommodations. To prove the first element in a failure to accommodate claim, Gleed must claim he is disabled. What were Gleed's disabilities?...
-
Simplify each expression. -6(3i)(-2i) = 2(31)(-2 +41) =
-
Nick and Kate can pedal around the lake 4 times as fast as Tim and Amy. If Nick and Kate go around the lake 52 times before stopping, how many more laps around the lake do Tim and Amy need to do...
-
Comfy Home employs part-time staff in its stores to help them with customers. Staff are paid an hourly wage and do not receive benefits. Between March 1st and 20th, the staff worked 400 hours at $15...
-
A call option with a strike price of $55 can be bought for $4. What will be your net profit if you sell the call and the stock price is $52 when the call expires? (2) If a corporation pays a dividend...
-
Classify the following accounts into one of the following categories: a. Assets b. Liabilities c. Equity d. Revenue e. Expense f. Off-balance-sheet activities (1) Service fees charged on deposit...
-
What types of inventory issues Starbucks might reflect upon at the end of each year? The mission of Starbucks is to inspire and nurture the human spiritone person, one cup, and one neighborhood at a...
-
Use MATLAB to find the following product: (10x 3 - 9x 2 - 6x + 12)(5x 3 - 4x 2 - 12x + 8)
-
Use MATLAB to find the quotient and remainder of 143 ? + 3 + 9 5x? + 7x 4
-
Use MATLAB to evaluate at x = 5. 24x 9x2 - 7 10x + 5x - 3x 7
-
A, B, C and D allegedly rob a Town Bank, which is located in the city of Cape Town. Members of the South African Police Services (SAPS) are called to the scene of the crime, during which a shoot-out...
-
Need help expanding the questions below. State of Ohio's position on the ban? What legislation if any supports or opposes the ban and or tries to protect smokers in the workplace? How smoking in...
-
A ray of light is traveling from water (n = 1.33) into an unknown substance. The angle of incidence at the boundary is 35 degrees and the refracted angle is 52 degrees. A) Calculate the index of...

Study smarter with the SolutionInn App


