Creatine C 4 H 9 N 3 O 2 has been used by body builders and athletes
Question:
Creatine C4H9N3O2 has been used by body builders and athletes to increase muscle mass and strength. Its chemical structure (not showing all of its carbon atoms and attached hydrogens) is
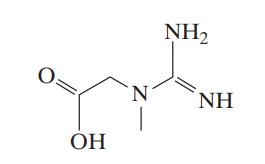
The following values are known for its dissociation constants:
![]()
a. Determine the charge on this amino acid as a function of pH.
b. Find its isoelectric point.
Transcribed Image Text:
0. OH N NH₂ ΝΗ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a To determine the charge on creatine as a function of pH we need to look at the dissociation consta...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The compound levodopa C 9 H 11 NO 4 (usually referred to a L-dopa) is used in the treatment of Parkinsons disease. The chemical structure (not showing its carbon atoms and attached hydrogens) is The...
-
The following values are known for the amino acid serine C 3 H 7 NO 2 S: Determine the charge on this amino acid as a function of pH. How does your result compare with the reported isoelectric point...
-
This is a legal memorandum that determines whether or not our client, John Rollins, has had his First Amendment rights violated. The operative facts are as follows. In 2009, Rollins was convicted of...
-
In Exercises use a graphing utility to graph the curve represented by the parametric equations. Indicate the direction of the curve. Identify any points at which the curve is not smooth. Curtate...
-
Why is the dual-rate charging method better than the single-rate method? In what circumstances would it not matter whether dual or single rates were used?
-
The Commercial Division of LaSalle Company has income from operations of $135,000 and assets of $650,000. The minimum acceptable rate of return on assets is 10%. What is the residual income for the...
-
Draw a cash flow diagram of any investment that exhibits both of the following properties: 1. The investment has a 4-year life. 2. The investment has a 10 percent/year internal rate of return.
-
The common stock of Warner Inc. is currently selling at $110 per share. The directors wish to reduce the share price and increase share volume prior to a new issue the per share par value is $10;...
-
A bank in Toronto charges 2.2% commission to buy and sell currencies. Assume that the current exchange rate is US$1 = C$1.1153. a. How many Canadian dollars will you have to pay to purchase US$4,500?...
-
Derive the equation that replaces Eq. 13.6-3 for a protein that has six ionizable sites. Macid = MHA + MHA- + MA_ = MHA + K MHA 1+ [ = K MH+ + K (MH+)] . MHA. K MHA K K + MH+ (MH+) . (13.6-3)
-
Oxalic acid C 2 H 2 O 4 is a dibasic acid with the following values for its ionization constants: Determine the charge on oxalic acid as a function of pH. pK = 1.2 and pK = 4.2
-
Identify the superkeys, candidate key(s), and the primary key for the following relation instance of the STU-CLASS relation schema. Student Number Student ACCOUNTING STEDRY ANTHROPOLOGY AP150 MWE9 5...
-
What is the difference between an American and European option? Is an American call option on a dividend-paying stock always worth at least as much as its intrinsic value? How about for a European...
-
In 2012 News Corp was rumoured to be considering selling off its newspaper line. What is the benefit of a spin-off of this type? Why would another company buy the assets?
-
What are the costs to a company of financial distress? Discuss your answer using a case study company of your choice.
-
During the financial crisis that engulfed most of Europe, two large banks, Lloyds TSB Group and HBOS, merged with each other to diversify risk. Is this a good or bad idea? Explain.
-
Given that many multinationals based in many countries have much greater sales outside their domestic markets than within them, what is the particular relevance of their domestic currency?
-
Enviro Company issues 8%, 10-year bonds with a par value of $ 250,000 and semiannual interest payments. On the issue date, the annual market rate for these bonds is 10%, which implies a selling price...
-
Arlington Merchants reported the following on its income statement for the fiscal years ending December 31, 2016 and 2015. 2016 2015 Sales $4,857,500 $4,752,900 Cost of goods sold 3,258,950 3,207,000...
-
The belt on the portable dryer wraps around the drum D, idler pulley A, and motor pulley B.If the motor can develop a maximum torque of M = 0.80 Nm, determine the smallest spring tension required to...
-
The 20-kg motor has a center of gravity at G and is pinconnected at C to maintain a tension in the drive belt. Determine the smallest counterclockwise twist or torque M that must be supplied by the...
-
The collar bearing uniformly supports an axial force of P = 5 kN. If the coefficient of static friction is s = 0.3, determine the smallest torque M required to overcome friction. M 150 mm- - 200 mm -
-
District Attorneys have decided not to prosecute drug and property crimes. Do you believe this policy is good or bad for criminal justice agencies? Please explain in details.
-
Subheading Elements and Performance Criteria ELEMENT PERFORMANCE CRITERIA Elements describe the essential outcomes. Performance criteria describe the performance needed to demonstrate achievement of...
-
MANUFACTURING. Kelly Industries manufactures two different structural support products used in the construction of large boats and ships. The two products, the Z345 and the W250, are produced from...

Study smarter with the SolutionInn App


