Estimate the triple-point temperature and pressure of benzene. The following data are available: T (C) pvap (Pa)
Question:
Estimate the triple-point temperature and pressure of benzene. The following data are available:
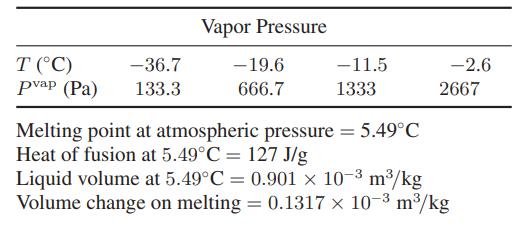
Transcribed Image Text:
T (°C) pvap (Pa) -36.7 133.3 Vapor Pressure - 19.6 666.7 -11.5 1333 Melting point at atmospheric pressure = 5.49°C Heat of fusion at 5.49°C = 127 J/g -2.6 2667 Liquid volume at 5.49°C = 0.901 x 10-³ m³/kg Volume change on melting = 0.1317 x 10-³ m³/kg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The triple point of benzene is the temperature and pressure at which all three phases of benzene sol...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The temperature dependence of the vapour pressure of solid sulfur dioxide can be approximately represented by the relation log (p/Torr) = 10.5916 - 1871.2/ (T/K) and that of liquid sulfur dioxide by...
-
A reasonable approximation to the vapor pressure of krypton is given by log 10 (P/Torr) = b 0.05223(a/T). For solid krypton, a = 10065 K and b = 7.1770. For liquid krypton, a = 9377.0 K and b =...
-
Andrew Coal Mining, Inc. is considering opening a strip mine, the cost of which is $8.8 million. Cash flows will be $55.4 million, all coming at the end of one year. The land must be returned to its...
-
In the real number system the equation a2 = 1 has exactly two solutions. Find at least eight different 3 3 matrices that satisfy the equation A2 = I3.
-
Girard & Company, Certified Public Accountants, audited the financial statements included in the annual report submitted by PMG Enterprises, Inc., to the Securities and Exchange Commission (SEC). The...
-
Isothermal compressibility of a substance is given by (a) \(\alpha=\frac{1}{V}\left(\frac{\partial V}{\partial P} ight)_{T}\) (b) \(\alpha=-\frac{1}{V}\left(\frac{\partial V}{\partial P} ight)_{T}\)...
-
Two springs, having different stiffnesses \(k_{1}\) and \(k_{2}\) with \(k_{2}>k_{1}\), are placed on either side of a mass \(m\), as shown in Fig. 13.27. When the mass is in its equilibrium...
-
George Zidek Corporation bought a machine on June 1, 2012, for $31,000, f.o.b. the place of manufacture. Freight to the point where it was set up was $200, and $500 was expended to install it. The...
-
You need to accumulate $10,000. To do so, you plan to make deposits of $1,400 per year - with the first payment being made a year from today - into a bank account that pays 7% annual interest. Your...
-
To answer the following questions, use Boeing Companys financial statements available for download from McGraw-Hills Connect or your course instructor (see the Preface for more information). a. For...
-
A thermally insulated (adiabatic) constant-volume bomb has been very carefully prepared so that half its volume is filled with water vapor and half with subcooled liquid water, both at 10C and 0.2876...
-
The vapor pressure of some materials can be represented by the equation Values of the constants in this equation are given below. Compute the heats of sublimation of the solids and the heats of...
-
Dudley Company failed to recognize the following accruals. It also recorded the prepaid expenses and unearned revenues as expenses and revenues, respectively, in the following year when paid or...
-
Prepare a good e-mail to all employees in the organization regarding the company's new accident insurance program. Some details you good include: The company has recently gone public with its shares....
-
Write a blog post of approximately 400 words discussing how technologies play a role in advancing communication integration in the organisation by referring to aligning messages across channels.
-
What effects do you think positive and negative thoughts, images, and self-talk have on performance? Share an example of a time when positive or negative thoughts/self-talk have affected your...
-
Knowing that the program's department head is not technology-savvy, what steps would you take to get these new tablets into the classroom? Describe at least 3 steps you would take to reach your goal...
-
A +2.0-nC point charge is placed at one corner of a square (1.5 m on a side), and a-3.0-nC charge is placed on a corner diagonally away from the first charge. What is the magnitude of the electric...
-
A bank is considering two securities: a 30-year Treasury bond yielding 7 percent and a 30-year municipal bond yielding 5 percent. If the banks tax rate is 30 percent, which bond offers the higher tax...
-
Suppose that you could invest in the following projects but have only $30,000 to invest. How would you make your decision and which projects would you invest in? Project Cost $ 8,000 11,000 9,000...
-
The geartrain in the transmission for the Segway Personal Transporter uses helical gears to reduce noise and vibration (Figure P8.28). The vehicles wheels have a diameter of 48 cm, and it has a top...
-
The mechanism that operates the load/unload tray for holding the disk in a tabletop Blu-rayTM player uses nylon spur gears, a rack, and a belt drive (Figure P8.29). The gear that meshes with the rack...
-
Explain why the number of teeth on the ring gear in a planetary geartrain is related to the sizes of the sun and planet gears by the equation N r = N s + 2N p .
-
Shankar Company uses a perpetual system to record inventory transactions. The company purchases inventory on account on February 2 for $35,000 and then sells this inventory on account on March 17 for...
-
Discuss a literature review for the topic " managing health and safety in the military" . Include citation with a short explication.
-
Problem 4: The 10 kg rod is released from rest when 0=0. The spring is unstretched when 0 = 0. Find the angular velocity of the rod when 0 = 30. The mass of the spring is 1 kg. O 1.5 m 2 m k = 80 N/m

Study smarter with the SolutionInn App


