Balance the following equations using the smallest whole number coefficients, then write the expression for K for
Question:
Balance the following equations using the smallest whole number coefficients, then write the expression for K for each reaction: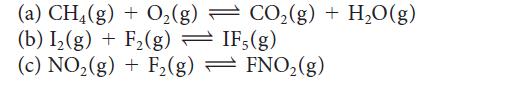
Transcribed Image Text:
(a) CH4(g) + O₂(g) (b) I₂(g) + F₂(g) → IF, (g) (c) NO₂(g) + F₂(g) = FNO₂(g) CO₂(g) + H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a CH4g 2Og COg 2 H0g ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations using the smallest whole number coefficients, then write the expression for K for each reaction: (a) CH4(g) + Cl(g) CHCl(g) + HCl(g) (b) NH3(g) + ClF3(g) (c) N(g) +...
-
1. Given Pascal's Triangle 4 1 3 1 2 6 1 3 1 5 10 10 4 1 5 1 1 a. Explain how the n and r values for C, relate to the number 6 shown above in Pascal's triangle. Justify your explanation by showing...
-
Balance the following equations using the method outlined in Section 3.7: (a) C + O2 CO (b) CO + O2 CO2 (c) C1 + Br2 HBr (d) K + C1O KOH + C1 (e) Mg + O2 MgO (f) O3 O2 (g) C1O2 C1O + O2 (h) N2 + C1...
-
Suppose that today, you paid $1,000 for the bond described in Problem 8 of Chapter 8, The net present value functions: NPV and XNPV. What would be the bonds IRR? A bonds IRR is often called the yield...
-
Isopropanol, containing 13 wt% water, can be dehydrated to obtain almost pure isopropanol at a 90% recovery by azeotropic distillation with benzene. When condensed, the overhead vapor from the column...
-
You are auditing a bank, and someone provides you with an anonymous tip that an employee is embezzling money from the bank. You decide to investigate the allegation. Your interviews with other bank...
-
Acetone is evaporating in a mixture of nitrogen and helium. Find the rate of evaporation and compare it with the rates in pure nitrogen and pure helium. Also compare it with the model using a...
-
Multiple Choice Questions Select the best answer for each of the following and explain fully the reason for your selection. a. Which of the following is least likely to be among the auditors...
-
A scientific instrument that accurately measures the strength of adhesives (sticky tape). -Tape Adhesion strength testing . How it works: The stepper motor drives the probe vertically down and...
-
At the beginning of the current period, Coe Ltd. had balances in Accounts Receivable of 200,000 and in Allowance for Doubtful Accounts of 9,000 (credit). During the period, it had net credit sales of...
-
You have two beakers: one is filled with tetrachloromethane and the other with water. You also have two compounds, butane(CH 3 CH 2 CH 2 CH 3 ) and calcium chloride. (a) In which liquid will butane...
-
A reactor for the production of ammonia by the Haber process is found to be at equilibrium with P N2 = 3.11 bar, P H2 = 1.64 bar, and P NH3 = 23.72 bar. If the partial pressure of N 2 is increased by...
-
Indicate which of the following is an example of a sample with replacement and which is a sample without replacement. a. Five friends go to a livery stable and select five horses to ride (each friend...
-
LeadersAngle Gene Deszca Organisational Change14:59 minutes https://www.youtube.com/watch?v=n9lzudH-uJI Evaluate yourself on the core competencies mentioned in the video. What do you think that you...
-
Calculate the break-even level of output.
-
Determine an investment's payback period.
-
Compute rates of return.
-
The following data relate to the characteristics of high-definition televisions A through E. Identify the individuals, variables, and data corresponding to the variables. Determine whether each...
-
What uniform series of cash flows is equivalent to a $100,000 cash flow, fifteen years from now, if the uniform cash flows occur at the end of the year for the next fifteen years and the periodic...
-
ABC company leased new advanced computer equipment to STU Ltd on 1 January 2019.STULtd has to pay annual rental of $290,000 starting at 1 January 2019. It is a four years lease with ultimate rental...
-
Compare the structure of vitamin E with the structures of BHT and BHA, and then determine which hydrogen atom is most easily abstracted from vitamin E.
-
Using your results from Problems P5.18 and P5.7, calculate ÎS, ÎS surroundings , and ÎS total for each step in the cycle and for the total Carnot cycle described in Figure 5.2....
-
Predict the products for each reaction. In each case, be sure to consider whether a chirality center is being generated and then draw all expected stereoisomers. (a) (b) (c) (d) (e) (f) H ROOR HBr...
-
K A universal set U consists of 16 elements. If sets A, B, and C are proper subsets of U and n(U) 16, n(An B) = n(An C) = n(BNC) = 6, n(AnBn C) = 2, and n(A UBU C) = 14, determine each of the...
-
Without solving, say whether the equation has two solutions, one solution, or no solution. The equation has because (x+4) = 19
-
4. Find the basis of R2 in which coordinates of the vector v = [2,-5] are (3, 1). 5. Find the basis of R3 in which coordinates of the vector v = [1,6,-4] are (1, 2, 4).

Study smarter with the SolutionInn App


