Below are ball-and-stick models of two molecules. In each case, indicate whether there must be, may be,
Question:
Below are ball-and-stick models of two molecules. In each case, indicate whether there must be, may be, or cannot be one or more lone pairs of electrons on the central atom.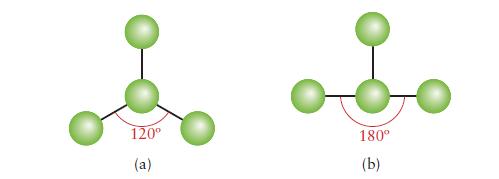
Transcribed Image Text:
120° (a) 180° (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
X 120 each M X b Explanation a structure where the central at...View the full answer

Answered By

Bree Normandin
Success in writing necessitates a commitment to grammatical excellence, a profound knack to pursue information, and a staunch adherence to deadlines, and the requirements of the individual publication. My background comprises writing research projects, research meta-analyses, literature reviews, white paper reports, multimedia projects, reports for peer-reviewed journals, among others. I work efficiently, with ease and deliver high-quality outputs within the stipulated deadline. I am proficient in APA, MLA, and Harvard referencing styles. I have good taste in writing and reading. I understand that this is a long standing and coupled with excellent research skills, analysis, well-articulated expressions, teamwork, availability all summed up by patience and passion. I put primacy on client satisfaction to gain loyalty, and trust for future projects. As a detail-oriented researcher with extensive experience surpassing eight years crafting high-quality custom written essays and numerous academic publications, I am confident that I could considerably exceed your expectations for the role of a freelance academic writer.
5.00+
7+ Reviews
21+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Below are ball-and-stick models of two molecules. In each case, indicate whether there must be, may be, or cannot be one or more lone pairs of electrons on the central atom. 120 (a) 180 (b)
-
What is the amount of a loss that an insured person agrees to bear personally? _______ is the amount of a loss that an insured person agrees to bear personally. What is the effect in the auto...
-
Draw the Lewis structure for SeS 3 and answer the following questions. How many valence electrons are present in this compound? How many bonding electrons are present in this compound? How many lone...
-
What conditions must be met for revenue to be recorded? Can pledges meet those conditions?
-
Jewett Online Company has the following liability accounts after posting adjusting entries: Accounts Payable $63,000, Unearned Ticket Revenue $24,000, Estimated Warranty Liability $18,000, Interest...
-
Find t such that -/2 t /2 and t satisfies the stated condition. sin t = sin(3/4)
-
In May 2010, Coquina Investments, an investment firm based in Corpus Christi, Texas, brought a lawsuit against TD Bank alleging that through the fraudulent actions of its agents, TD Bank is...
-
(Assumptions, Principles, and Constraints) Presented below are the assumptions, principles, and constraints used in this chapter. 1. Economic entity assumption 5. Historical cost principle 9....
-
(a) Identify and explain the various ways through which government can intervene and assist upcoming Small entrepreneurs to grow and sustain their enterprises. (b) Explain the rationale behind SWOT...
-
The sulfate ion, SO 4 2 , is present in a number of important minerals, including gypsum (CaSO 4 2H 2 O), which is used in cement, and Epsom salts (MgSO 4 7H 2 O), which is used as a purgative....
-
Draw the Lewis structure of (a) CCl 4 ; (b) COCl 2 ; (c) ONF; (d) NF 3 .
-
Without referring to tables, predict which of the following has the higher enthalpy in each case: (a) 1 mol CO2(s) or 1 mol CO2(g) at the same temperature, (b) 2 mol of hydrogen atoms or 1 mol of H2...
-
Define virtualization. How does it relate to the cloud?
-
Why should the data access and manipulation classes be dependent on the problem domain classes instead of the other way around?
-
Why is Parkinsons Law relevant to the design of the physical architecture layer?
-
What are the obstacles for provisioning the physical architecture layer with cloud technologies?
-
A Cobb-Douglas production function is Q = 12L.6K.5. What happens to Q if L increases by 1%? If K increases by 1%? What can you say about returns to scale?
-
A vertical spring with a spring constant of 450 N/m is mounted on the floor. From directly above the spring, which is unstrained, a 0.30-kg block is dropped from rest. It collides with and sticks to...
-
During the month, services performed for customers on account amounted to $7,500 and collections from customers in payment of their accounts totaled $6,000. At the end of the month, the Accounts...
-
A 230.-mL sample of a 0.275 M CaCl 2 solution is left on a hot plate overnight; the following morning, the solution is 1.10 M. What volume of water evaporated from the 0.275 M CaCl 2 solution?
-
A 50.00-mL sample of a solution containing Fe 2+ ions is titrated with a 0.0216 M KMnO 4 solution. It required 20.62 mL of KMnO4 solution to oxidize all the Fe 3+ ions to Fe 3+ ions by the reaction
-
A 50.00-mL sample of a solution containing Fe 2+ ions is titrated with a 0.0216 M KMnO 4 solution. It required 20.62 mL of KMnO4 solution to oxidize all the Fe 3+ ions to Fe 3+ ions by the reaction
-
You are offered an investment opportunity that will pay you $15,000 per year at the beginning of each of the next 10 years. The investment will cost you $100,000 today. Based on this information,...
-
calculate the economic value added of the hardware division based in the following additional information weightage average cost 9%, current liabilities$2000000 and tax rate 30%. briefly comment on...
-
You purchase a house for $120,864.00. You made a down payment of 20,000 and the remainder of the purchase price was financed with a mortgage loan. The mortgage loan is a 30-year mortgage with an...

Study smarter with the SolutionInn App


