Question: Below are two absorption plots. One was obtained from a solution of an organic dye and the other from a quantum dot suspension. Which plot
Below are two absorption plots. One was obtained from a solution of an organic dye and the other from a quantum dot suspension. Which plot was obtained from which solution? Explain your reasoning.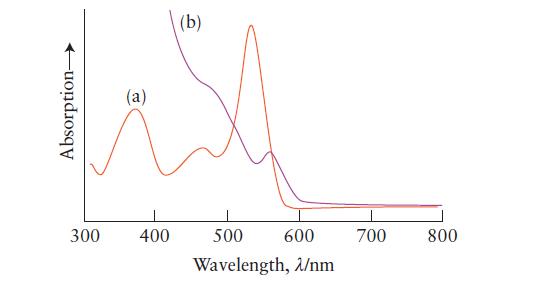
Absorption (a) 300 400 (b) 500 600 Wavelength, 2/nm 700 800
Step by Step Solution
3.37 Rating (169 Votes )
There are 3 Steps involved in it
Plot a Was obtaine... View full answer

Get step-by-step solutions from verified subject matter experts


