Below is pictured the reaction between an atom of magnesium and an atom of oxygen. Identify each
Question:
Below is pictured the reaction between an atom of magnesium and an atom of oxygen. Identify each element and the ions formed and explain your reasoning.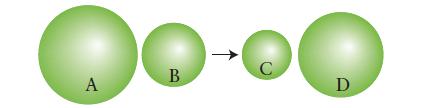
Transcribed Image Text:
A B C D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Element A represents magnesium atom Mg and element B represents oxygen atom O Magnesium Mg has ...View the full answer

Answered By

Charles Okinda
students should give all the instructions concerning the challenge that they face. they will get an immediate response because I am always online.
4.90+
753+ Reviews
1480+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Below is pictured the reaction between an atom of sodium and an atom of chlorine. Identify each element and the ions formed and explain your reasoning. A D D
-
At a temperature of 35 F, the cylinder shown has an outer diameter of 3in, a wall thickness of 1/8 in, and length of 2.5 ft. It has a fixed support at its base and its free end is 1/128in from the...
-
Consider the reaction between oxygen (O 2 ) gas and magnesium metal to form magnesium oxide. Using oxidation states, how many electrons would each oxygen atom gain, and how many electrons would each...
-
The 60-mm-diameter shaft rotates at 300 rev/min. This motion is caused by the unequal belt tensions on the pulley of 800 N and 450 N. Determine the power transmitted and the maximum shear stress...
-
The chandelier is supported by three chains which are concurrent at point O. If the resultant force at O has a magnitude of 130 lb and is directed along the negative z axis, determine the force in...
-
In Exercises use integration tables to find the indefinite integral. et (1-ex)3/2 dx
-
You need to use a long solenoid to produce a magnetic field of magnitude \(0.070 \mathrm{~T}\). If the maximum current you are able to run through the windings is \(20 \mathrm{~A}\), what is the...
-
A computer problem at Southglenn Photography Studios has resulted in incomplete financial statements. Management of the company has asked you to see if you can fill in the missing data. a. Recreate...
-
1. Where cursor implementation can be used? 2. List down the applications of List. 3. What are the advantages of linked list? 4. Mention the demerits of linked list? 5. What are the operations...
-
Neon lights glow with orange light and they also emit radiation of wavelength 865 nm. Calculate the energy change resulting from the emission of 1.00 mol of photons at this wavelength.
-
For each of the following ground-state atoms, predict the type of orbital (1s, 2p, 3d, 4f, etc.) from which an electron will be removed to form the 1 1 ion: (a) Zn; (b) Cl; (c) Al; (d) Cu.
-
Suppose all the alpha particles in Rutherfords experiment went straight through the gold foil with absolutely no deflections. What would this imply about the structure of the atom?
-
Matrix multiplication is a key operation supported in hardware by the TPU. Before going into details of the TPU hardware, its worth analyzing the matrix multiplication calculation itself. One common...
-
Figure 6.1 is a listing of outages in an array of servers. When dealing with the large scale of WSCs, it is important to balance cluster design and software architectures to achieve the required...
-
Increasing a cache's associativity (with all other parameters kept constant) statistically reduces the miss rate. However, there can be pathological cases where increasing a cache's associativity...
-
Based on the observations in Figures 6.12 and 6.13, what can you say qualitatively about the trade-offs between revenue loss from downtime and costs incurred for uptime? Figure 6.12: Figure 6.13:...
-
When a cloud computing service provider receives jobs consisting of multiple Virtual Machines (VMs) (e.g., a MapReduce job), many scheduling options exist. The VMs can be scheduled in a round-robin...
-
Yount Company reports the following for the month of June. Instructions (a) Compute the cost of the ending inventory and (he cost of goods sold under (I) FIFO (2) Average-cost. (b) Which costing...
-
In your audit of Garza Company, you find that a physical inventory on December 31, 2012, showed merchandise with a cost of $441,000 was on hand at that date. You also discover the following items...
-
The complex ion PdCL42- is diamagnetic. Propose a struc-ture for PdCL42-.
-
Why are CN- and CO toxic to humans?
-
In the production of printed circuit boards for the elec¬tronics industry, a 0.60-mm layer of copper is laminated onto an insulating plastic board. Next, a circuit pattern made of a chemically...
-
In this question we consider a simplified two-dimensional cellular automaton model for the growth of a tumour in healthy tissue. The alphabet is (H, A,Q, N) where I denotes a healthy cell, A denotes...
-
A firm that is currently locating facilities in a large number of other countries in order to capitalize on lower production and distribution costs is at what level of global participation?...
-
B of Bombay consigned 400 packages of coffee to K of Kanpur. The cost of each package was Rs. 300. A sum of Rs. 2,000 was paid towards freight and insurance by B. In the transit, 60 packages were...

Study smarter with the SolutionInn App


