Calculate the energy released per gram of starting material in the nuclear reaction represented by each of
Question:
Calculate the energy released per gram of starting material in the nuclear reaction represented by each of the following equations: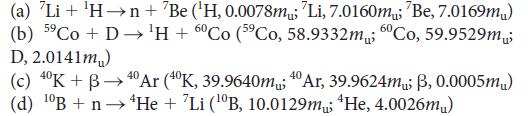
Transcribed Image Text:
(a) 'Li + Hn + Be (H, 0.0078m; 'Li, 7.0160m; 'Be, 7.0169m) (b) 59 Co + D H + 60 Co (59Co, 58.9332m; 60Co, 59.9529m; D, 2.0141m) (c) 40K + B40 Ar (40K, 39.9640m 40Ar, 39.9624m; B, 0.0005m) (d) 0B + n He + Li (0B, 10.0129m He, 4.0026m)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The energy released in a nuclear reaction can be calculated using the mass defect and Einsteins equation Emc2 where E is the energy m is the mass lost ...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Calculate the energy released per gram of starting material in the fusion reaction represented by each of the following equations: (a) D DHe + n (D, 2.0141m; 'He, 3.0160m) (b) He + D He + H (H,...
-
Comparison of Energy Released per Gram of Fuel. (a) When gasoline is burned, it releases 1.3 x 108 J of energy per gallon (3.788 L). Given that the density of gasoline is 737kg/m3, express the...
-
Calculate the amount of energy released per gram of hydrogen nuclei reacted for the following reaction. The atomic masses are 11H, 1.00782 amu, and 21H, 2.01410 amu. 11H + 11H 21H +Je
-
Fill in the missing amounts for the following bank reconciliation: Practice Exercise3 Bank Reconciliation March 31,20 Bank Statement Balance $3,764.00 Add: Deposit in transit $4,031.00 Deduct:...
-
Use the differential equation approach to find i(t) for t > 0 in the circuit in figure and plot the response including the time interval just prior to opening the switch. 2 kn i(t) for 5 kn 1 kn 2 kn...
-
Differentiate between data redundancy and concurrency.
-
Find the values of the 5 -month call option of Example 19.1 using the same trinomial lattice used in that example but employing the utility function \(U(x)=\sqrt{x}\). What is \(\alpha\) ? Example...
-
On January 1, 2012, Witzling Company issued $4,000,000 face value, 8%, 15-year bonds at $3,391,514. This price resulted in an effective-interest rate of 10% on the bonds. Witzling uses the...
-
1. Construct a truth table for the following: a. out x'yz +(xyz)' b. out (x+z')(x+y')(y+z) 2. Simplify the following functional expressions using Boolean algebra and its identities. List the identity...
-
(a) What percentage of a strontium-90 sample remains after 8.5 a? (b) Determine the percentage of an iodine-131 sample that remains after 6.0 d.
-
It is found that 20. mmol 222 Rn (t 1/2 = 3.82 d) has seeped into a closed basement with a volume of 2.0 3 103 m3. (a) What is the initial activity of the radon in picocuries per liter (pCi L 1 )?...
-
Journalise the following transactions assuming cost and financial accounts are integrated: Raw materials purchases Direct materials issued to production Wages paid (30% indirect) Direct wages charged...
-
Problem 2 Below is three months of data on total sales from McDonalds in Huntsville. Using the data below, please answer the following question. Month August September October Sales $30,000 $45,000...
-
What were some of the primary constraints for this project? How did quality issues in the design impact manufacturing and force a difficult schedule tradeoff? Despite early enthusiasm, what were the...
-
choose an area relevant to your area of training in the Tourism, Hospitality and Events Industry. This may include Accommodation Services, Front Office, Cookery, Food and Beverage, Tourism or Events....
-
Students need to identify appropriate research design and methods to achieve the research objectives. Provide adequate references to the research studies which used or discussed the proposed research...
-
Name three functions of Canvas. 2. Is there a function of Canvas that you did not know about but will use in this course? 3. Will you be using the mobile Canvas App? 4. What do you want to learn in...
-
Suppose that a manager has a choice between the following two mathematical models of a given situation: (a) A relatively simple model that is a reasonable approximation of the real situation, (b) A...
-
a. Determine the domain and range of the following functions.b. Graph each function using a graphing utility. Be sure to experiment with the window and orientation to give the best perspective of the...
-
The 50-mm-diameter A992 steel shaft is subjected to the torques shown. Determine the angle of twist of the end A. 400 N-m 200 N-m 300 mm 800 N-m 600 mm 600 mm
-
The solid shaft has a linear taper from r A at one end to r B at the other. Derive an equation that gives the maximum shear stress in the shaft at a location x along the shafts axis.
-
The assembly shown consists of two A992 steel bolts AB and EF and an 6061-T6 aluminum rod CD. When the temperature is at 30° C, the gap between the rod and rigid member AE is 0.1 mm. Determine...
-
A law firm has accepted five new cases, each of which can be adequately handled by any one of its five junior partners. Due to differences in experience and expertise, however, the junior partners...
-
Your Company has 10 employees. Nine are paid $35,000 - $50,000 a year and one is paid $5,000. Compute Your Company's FUTA liability for 2023 $3,780 $300 $4,080 $2,400
-
Sasha is in a project meeting where a major risk was revealed to be impacting the project. Sashas boss is in another meeting for the rest of the day, but she needs to provide a detailed update to her...

Study smarter with the SolutionInn App


