Consider the following galvanic cell: What happens to (mathscr{E}) as the concentration of (mathrm{Zn}^{2+}) is increased? as
Question:
Consider the following galvanic cell:
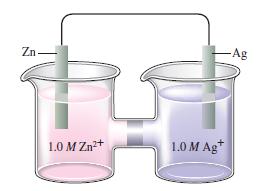
What happens to \(\mathscr{E}\) as the concentration of \(\mathrm{Zn}^{2+}\) is increased? as the concentration of \(\mathrm{Ag}^{+}\)is increased? What happens to \(\mathscr{E}^{\circ}\) in these cases?
Transcribed Image Text:
Zn 1.0 M Zn+ 1.0 M Ag+ -Ag
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
In order to examine the variations in both standard cell potential C and cell potential E E We may u...View the full answer

Answered By

Akash Goel
I am in the teaching field since 2008 when i was enrolled myself in chartered accountants course
Since then i have an experience of teaching of class XI, XII, BCOM, MCOM, MBA, CA CPT.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following galvanic cells: For each galvanic cell, give the balanced cell equation and determine Ïo. Standard reduction potentials are found in Table 11.1. Table 11.1 1.0 M Cut 1.0 M...
-
Consider the following galvanic cell at 25oC: Pt | Cr2+ (0.30 M), Cr3+(2.0 M) | | Co2+(0.20 M) | Co The overall reaction and equilibrium constant value are 2Cr2+(aq) + Co2+(aq) 2Cr3+(aq) + Co(s) K =...
-
Consider the following galvanic cell: Calculate the concentrations of Ag+(aq) and Ni2+(aq) once the cell is dead. Ni 1.0 MNi2+ 10 M Ag
-
Should U.S. national forests become privatized (sold to private owners)? Why or why not?
-
Include air resistance proportional to the square of the balls speed in the previous problem. Let the drag coefficient be cw = 0.5, the softball radius be 5 cm and the mass be 200 g. (a) Find the...
-
Water is in the pipe shown in Fig. 3.26. Calculate the pressure at point A in kPa(gage). Pipe 100 mm - Water 75 mm Mercury (sg = 13.54)
-
North Shore Architectural Stone, Inc., a company that installs limestone in residential and commercial buildings, agreed to supply and install limestone for a property owned by Joseph Vitacco. North...
-
Accounting for Self-Constructed Assets Troopers Medical Labs, Inc., began operations 5 years ago producing stetrics, a new type of instrument it hoped to sell to doctors, dentists, and hospitals. The...
-
Why is the relational model more important above the other models?
-
The saturated calomel electrode, abbreviated SCE, is often used as a reference electrode in making electrochemical measurements. The SCE is composed of mercury in contact with a saturated solution of...
-
What is the difference between \(\mathscr{E}\) and \(\mathscr{E}^{\circ}\) ? When is \(\mathscr{E}\) equal to zero? When is \(\mathscr{E}^{\circ}\) equal to zero? (Consider "regular" galvanic cells...
-
Let F(t, y) = t 2 y and let y(t) be the solution of dy/dt = F(t, y) satisfying y(2) = 3. Let h = 0.1 be the time step in Eulers Method, and set y 0 = y(2) = 3. (a) Calculate y 1 = y 0 + hF(2, 3)....
-
A company reports the following for the prior year: $ 1 . 6 million in sales $ 1 million in total assets $ 1 6 0 , 0 0 0 in net income $ 8 0 0 , 0 0 0 in equity ( at the beginning of this prior year...
-
Civil society is becoming increasingly knowledgeable about governmental policies and actions. This is due to technological developments making occurrences almost instantaneously available to civil...
-
On January 1, 2023, Vin Diesel Inc purchases a fleet of six vehicles for $50,000 each. They pay cash for the purchase. Vehicles have zero salvage value for depreciation calculations. Vin Diesel...
-
Briefly discuss about the Financials ( Balance Sheet& Income Statement reports ) of the company. Why these report are important in supply chain decision making?
-
What Stock Control report can be used to review all inventory item information entered in the I/C Items form?
-
Refer to the data in Exercise 6-31. The president of Tiger Furnishings is confused about the differences in costs that result from using direct labor costs and machine-hours. Required a. Explain why...
-
A sprinkler head malfunctions at midfield in an NFL football field. The puddle of water forms a circular pattern around the sprinkler head with a radius in yards that grows as a function of time, in...
-
Calculate the equilibrium constants for the following reactions: (a) Mn(s) + Ti+ (aq) = (b) In+ (aq) + U+ (aq) U+ (aq) Mn+ (aq) + Ti(s) In+ (aq) + U4+ (aq)
-
You are an analytical chemist and are working out a procedure for precipitating lead from a solution of lead nitrate. You know that lead(II) iodide is insoluble, so you decide to use potassium iodide...
-
For reactions in which the equilibrium constant is very large or very small, it can be difficult to measure the concentration of all species in solution in order to determine K. An alternative method...
-
PROBLEM EXERCISE: 1. A power plant is said to have had a use factor of 48.5% and a capacity factor of 42.4%. How many hours did it operate during the year? (answer 7660 hrs) 2. A 50 000kw steam plant...
-
Consider flow through an annulus between two concentric horizontal cylinders (Fig. H7.4). The flow is driven by the inner cylinder, of radius ri, which moves axially with constant velocity, V, while...
-
For P7:- a. A lifting system is arranged using number N pulleys to lift a mass of m kg vertically through a height of h metres. It is found that a force of FN is needed to lift the load. Calculate:-...

Study smarter with the SolutionInn App


