Consider the hypothetical reaction, A 2 + B 2 2AB, where the rate law is: The
Question:
Consider the hypothetical reaction, A2 + B2 → 2AB, where the rate law is:
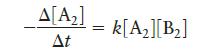
The value of the rate constant at 302°C is 2.45 × 10-4 L mol-1 s-1, and at 508°C the rate constant is 0.891 L mol-1 s-1. What is the activation energy for this reaction? What is the value of the rate constant for this reaction at 375°C?
Transcribed Image Text:
Δ[Α2] k[Ag][B2] ΔΕ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
a The activation energy for t...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the hypothetical reaction A + B + 2C 2D + 3E In a study of this reaction, three experiments were run at the same temperature. The rate is defined as 2d[B]/dt. Experiment 1: [A]0 = 2.0 M [B]0...
-
Consider the hypothetical reaction B E + F which is assumed to occur by the mechanism Where B* represents a B molecule with enough energy to surmount the reaction energy bar70. Consider the following...
-
Consider the hypothetical reaction A + B + 2C 2D + 3E where the rate law is An experiment is carried out where [A]0 = 1.0 Ã 10-2 M, [B]0 = 3.0 M, and [C]0 = 2.0 M. The reaction is started, and...
-
Suppose that in a particular area the consumption of water varies tremendously throughout the year, with average household summer use exceeding winter use by a great deal. What effect would this have...
-
The diameters of fully grown white oak trees are normally distributed, with a mean of 3.5 feet and a standard deviation of 0.2 foot, as shown in the figure. Random samples of size 16 are drawn from...
-
Given a 100-MHz clock signal, derive a circuit using D flip-flops to generate 50-MHz and 25-MHz clock signals. Draw a timing diagram for all three clock signals, assuming reasonable delays.
-
A long rotor can be balanced by adding weights in a. a single plane b. any two planes c. two specific planes
-
Hal Thomas, a 25-year-old college graduate, wishes to retire at age 65. To supplement other sources of retirement income, he can deposit $2,000 each year into a tax-deferred individual retirement...
-
In recent years, Sandhill Co. has purchased three machines. Because of frequent employee turnover in the accounting department, a different accountant was in charge of selecting the depreciation...
-
Go to the Webinars worksheet. DeShawn wants to determine the number of webinars the company can hold on Tuesdays and Thursdays to make the highest weekly profit without interfering with...
-
A certain substance, initially present at 0.0800 M, decomposes by zero-order kinetics with a rate constant of 2.50 102 -2 mol L -1 s - 1 . Calculate the time (in seconds) required for the system to...
-
The reaction A(aq) + B(aq) products(aq) was studied, and the following data were obtained: What is the order of the reaction with respect to A? What is the order of the reaction with respect to B?...
-
An isomorphism of a group with itself is an automorphism of the group. Find the number of automorphisms of the given group. Z
-
a. What is scenario analysis as applied to pricing decisions? b. Why is it such an important part of the process?
-
What is the role of accounting information in pricing decisions?
-
What elements of profit analysis change when a provider moves from a fee-for-service to a discounted fee-for-service environment?
-
a. Write out and explain the equation for volume breakeven. b. What role does contribution margin play in this equation?
-
What are its primary purposes and benefits?
-
A particular breed of dog can have long hair or short hair. When true-breeding long-haired animals were crossed to true-breeding short-haired animals, the offspring all had long hair. The F2...
-
Why did management adopt the new plan even though it provides a smaller expected number of exposures than the original plan recommended by the original linear programming model?
-
A 1.75 mole sample of an ideal gas is compressed isothermally from 62.0 L to 19.0 L using a constant external pressure of 2.80 atm. Calculate q, w, U, and H.
-
For each of the compounds below, locate the lone pair adjacent to a positive charge and draw the resonance structure: a. b. c. N.
-
Assume the following simplified dependence of the pressure in a ventricle of the human heart as a function of the volume of blood pumped. P s , the systolic pressure, is 120. mm Hg, corresponding to...
-
How would each leadership style ( transactional leadership and transformational leadership) approach communication with various stakeholders?
-
Explained the different type of leader -Great man -trait theory -skill theory -behavior theory -Contingency theory (situational) -Transformation transactional leadership -transactional leadership...
-
What effect will a corporation's emphasis on environmental responsibility have on its financial performance in both the short term and long term? Does this contradict the notation of maximizing the...

Study smarter with the SolutionInn App


