Determine which of the following compounds are stable with respect to decomposition into their elements under standard
Question:
Determine which of the following compounds are stable with respect to decomposition into their elements under standard conditions at 25°C (see Appendix 2A):
(a) PCl5(g);
(b) HCN(g);
(c) NO(g);
(d) SO2(g).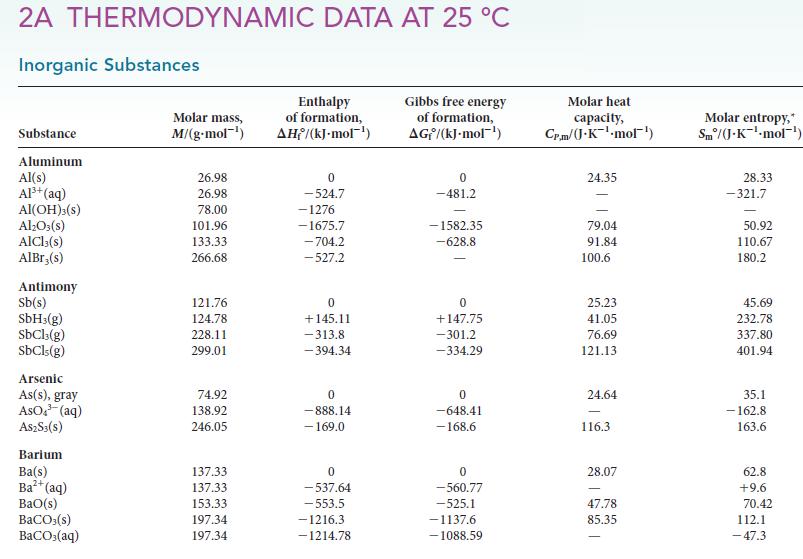
Transcribed Image Text:
2A THERMODYNAMIC DATA AT 25 °C Inorganic Substances Substance Aluminum Al(s) Al³+ (aq) AI(OH)3(s) Al₂O3(s) AlCl3(s) AlBr,(s) Antimony Sb(s) SbH3(g) SbCl3(g) SbCls (g) Arsenic As(s), gray AsO³(aq) A$2S3(S) Barium Ba(s) Ba²+ (aq) BaO(s) BaCO3(s) BaCO3(aq) Molar mass, M/(g.mol-¹) 26.98 26.98 78.00 101.96 133.33 266.68 121.76 124.78 228.11 299.01 74.92 138.92 246.05 137.33 137.33 153.33 197.34 197.34 Enthalpy of formation, AH/(kJ-mol-¹) 0 -524.7 -1276 -1675.7 -704.2 -527.2 0 +145.11 -313.8 -394.34 0 -888.14 - 169.0 0 -537.64 -553.5 -1216.3 -1214.78 Gibbs free energy of formation, AG/(kJ.mol-¹) 0 -481.2 -1582.35 -628.8 0 +147.75 -301.2 -334.29 0 -648.41 -168.6 0 -560.77 -525.1 -1137.6 -1088.59 Molar heat capacity, Cr.m/(J.K¹-mol¹) 24.35 79.04 91.84 100.6 25.23 41.05 76.69 121.13 24.64 116.3 28.07 47.78 85.35 Molar entropy,* Sm/(J-K¹-mol-¹) 28.33 -321.7 50.92 110.67 180.2 45.69 232.78 337.80 401.94 35.1 -162.8 163.6 62.8 +9.6 70.42 112.1 -47.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a and d ...View the full answer

Answered By

Ashwani Prasad
I had pursued B.E. in Chemical Engineering in 2018. I have worked as a Junior Engineer in the chemical industry. So, I know theoretical as well as the practical aspect of an engineering problem. I am keenly interested in Chemical process calculations, chemical reaction engineering and chemical technology.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Determine which of the following compounds are stable with respect to decomposition into their elements under standard conditions at 25C (see Appendix 2A): (a) C 3 H 6 (g), cyclopropane; (b) CaO(s);...
-
The term thermodynamic stability refers to the sign of r G . If r G is negative, the compound is stable with respect to decomposition into its elements. Use the data in Appendix D to determine...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Kaspar Corporation makes a commercial-grade cooking griddle. The following information is available for Kaspar Corporation's anticipated annual volume of 25,600 units. Per Unit Total Direct materials...
-
A point charge q is brought to a position a distance d away from an infinite plane conductor held at zero potential. Using the method of images, find: (a) The surface-charge density induced on the...
-
Why is PlatelmagesID the primary key for the Platelmages table rather than LicensePlateNumber? An Access database from a hypothetical small island nation contains simulated license plate information...
-
Jupiter, with an inertia 317.83 times that of Earth, is at an average distance of \(7.784 \times 10^{11} \mathrm{~m}\) from the Sun. At what distance from the centre of the Sun is the center of mass...
-
The ledger of Paisley Corporation at December 31, 2014, after the books have been closed, contains the following stockholders equity accounts. Preferred Stock (8,000 shares issued) ..............$...
-
Which one of the metal atoms whose electronic structure given below forms a nitrate of the type M(NO3)2? 37 A. 2.8.0 B. 2.8.1 C. 2.8.2 D. 2.8.3
-
Draw the Lewis structure for the hypothetical molecule N 6 , consisting of a six-membered ring of nitrogen atoms. Using bond enthalpies, calculate the enthalpy of reaction for the decomposition of N...
-
The following picture shows a molecular visualization of a system undergoing a spontaneous change. Account for the spontaneity of the process in terms of the entropy changes in the system and the...
-
Determine the velocity of cart A if cylinder B has a downward velocity of 2 ft/sec at the instant illustrated. The two pulleys at C are pivoted independently. UB B Problem 2/210
-
At the current price level, producers supply $375 billion of final goods and services while consumers purchase $355 billion of final goods and services. The price level is: a. above equilibrium. b....
-
Which of the following will shift the aggregate supply curve to the right? a. A new networking technology increases productivity all over the economy. b. The price of oil rises substantially. c....
-
Label each of the following descriptions as being either an immediate-short-run aggregate supply curve, a short-run aggregate supply curve, or a long-run aggregate supply curve. b. A vertical line....
-
Which of the following will shift the aggregate demand curve to the left? a. The government reduces personal income taxes. b. Interest rates rise. c. The government raises corporate profit taxes. d....
-
Which of the following help to explain why the aggregate demand curve slopes downward? a. When the domestic price level rises, our goods and services become more expensive to foreigners. b. When...
-
A state highway safety agency must allocate its budget for the next fiscal year. A total funding of $32 million has been granted for reducing fatalities and property damage due to automobile...
-
A heat engine has a heat input of 3 Ã 104 Btu/h and a thermal efficiency of 40 percent. Calculate the power it will produce, in hp. Source 3 x 10 Btu/h 40% HE Sink
-
Reagents such as HCl, HBr, and HOH (H 2 O) can add across carboncarbon double and triple bonds, with H forming a bond to one of the carbon atoms in the multiple bond and Cl, Br, or OH forming a bond...
-
Alkenes and cycloalkanes are structural isomers of each other. Give an example of each using C 4 H 8 . Another common feature of alkenes and cycloalkanes is that both have restricted rotation about...
-
Alkenes and cycloalkanes are structural isomers of each other. Give an example of each using C 4 H 8 . Another common feature of alkenes and cycloalkanes is that both have restricted rotation about...
-
There are 10 kilometers between Toy Town and Candy Land. A train carrying toys leaves Toy Town for Candy Land traveling at a rate of 4km/h. At the same time, a train carrying candy leaves Candy Land...
-
4) (a) Calculate the pore pressure, the total stress, and the effective stress at the indicated points in the figures below. (b) Determine the head loss, the hydraulic gradient, and the discharge...
-
The Shirt Shop had the following transactions for T-shirts for Year 1, its first year of operations. January 20 April 21 July 25 September 19 Purchased 540 units @ $ 7 Purchased 340 units @ $9 =...

Study smarter with the SolutionInn App


