Each of the pictures below shows a molecular view of a system undergoing a change. In each
Question:
Each of the pictures below shows a molecular view of a system undergoing a change. In each case, indicate whether heat is absorbed or given off by the system and whether expansion work is done on or by the system. Predict the signs of q and w for the process.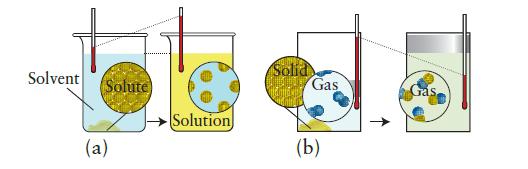
Transcribed Image Text:
Solvent Solute (a) Solution Solid Gas (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a A solid is dissolving in solution and as indicated there is no volu...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
340+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Each of the pictures below shows a molecular view of a system undergoing a change at constant temperature. In each case, indicate whether heat is absorbed or released by the system and whether...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Jinny Buffett recently retired as a flight attendant and is interested in opening a fitness center and health spa exclusively for women in Grand Cayman, where she resides. After careful study, she is...
-
For the year ended December 31, 2010, the job cost sheets of Moxie Company contained the following data. Other data:1. Raw materials inventory totaled $20,000 on January 1. During the year, $100,000...
-
Find the indicated vector sums and differences with the given vectors by means of diagrams. 2B 3/2E A B C D E
-
A \(15-\mathrm{kg}\) box is pushed at \(5 \mathrm{~m} / \mathrm{s}\) across a low-friction floor and is caught by a \(60-\mathrm{kg}\) man on low-friction skates, initially at rest. What is the...
-
1. Compare the first-digit frequency in the transactions in this table below with Benfords Law. What are the results? 2. Could fraud be occurring in thisorganization? AMOUNT DESCRIPTION CHECK NO...
-
A multinational corporation is accused of environmental pollution and human rights violations in a foreign country where it operates. What legal challenges arise in holding the corporation...
-
If you start with 276 g of liquid water at 25.C, how much heat must be supplied to convert all the liquid into vapor at 100.C?
-
Calculate the change in entropy when the pressure of 5.75 g of helium gas is decreased from 320.0 kPa to 40.0 kPa while the temperature decreases from 423 K to 273 K. Assume ideal behavior.
-
What is the difference between gap analysis and duration analysis? What is the purpose of gap analysis, and what is the purpose of duration analysis?
-
Disparate-treatment discrimination occurs when an employer intentionally discriminates against an employee. (True/False)
-
When is a shareholders appraisal right available?
-
Fast Jack is a fast-food restaurant that employs minors. Fast Jack is subject to the federal child labor, minimum wage, and overtime provisions in a. the Family and Medical Leave Act. b. the...
-
What are some protections afforded by the federal age-discrimination law?
-
National Supplies Company hires Linda and Brad as employees to deal with third-party purchasers and suppliers. Linda and Brad are a. principals. b. agents. c. both a and b. d. none of the above.
-
True or False: 1. A lower price will increase your consumer surplus for each of the units you were already consuming and will also increase your consumer surplus from increased purchases at the lower...
-
a. What is the cost of borrowing if Amarjit borrows $28 500 and repays it over a four-year period? b. How many shares of each stock would he get if he used the $28 500 and invested equally in all...
-
Which of the following statements is(are) true? Correct the false statement(s). a. When a reactant is added to a system at equilibrium at a given temperature, the reaction will shift right to...
-
Suppose the reaction system UO2(s) + 4HF(g) UF4(g) + 2H2O(g) has already reached equilibrium. Predict the effect that each of the following changes will have on the equilibrium position. Tell...
-
Consider the reaction: Fe3+(aq) + SCN2(aq) FeSCN2+(aq) How will the equilibrium position shift if a. Water is added, doubling the volume? b. AgNO3(aq) is added? (AgSCN is insoluble.) c. NaOH(aq) is...
-
Consider the following for Guardian Manufacturing Company: Change in finished goods inventory Change in work-in-process inventory Total manufacturing costs $ 315 increase $ 145 increase $ 630 What...
-
Cash Accounts Receivable Office Equipment Accumulated Depreciation Accounts Payable Capital Stock Retained Earnings Dividends Fees Earned Salaries Expense Advertising Expense Depreciation Expense...
-
Today ABC Corp. starts a project that requires it to invest in new equipment of $1,103,000. Doing so will earn ABC pre-tax cash flows of $158,000 annually over 10 years. The equipment has a salvage...

Study smarter with the SolutionInn App


