Estimate the density of each of the following solids from the ionic radii given in Fig. 1F.6:
Question:
Estimate the density of each of the following solids from the ionic radii given in Fig. 1F.6:
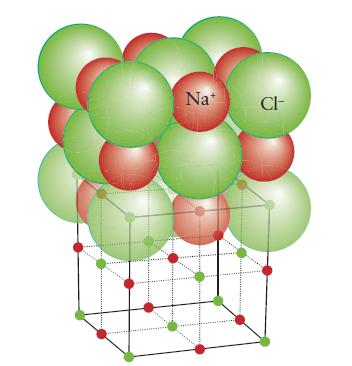
(a) Calcium oxide (rock-salt structure, Fig. 3H.28);
(b) Cesium bromide (cesium- chloride structure, Fig. 3H.30).
FIGURE 3H.30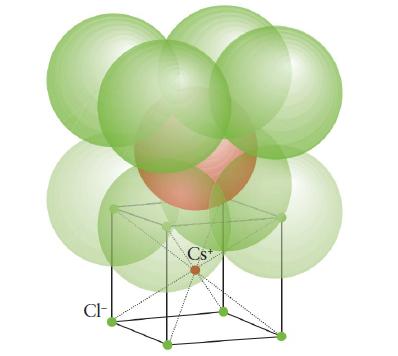
FIGURE 3H.28
Transcribed Image Text:
CI- Cs+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a d 3...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The table below lists the ionic radii for the cations and anions in three different ionic compounds. Each compound has either the NaCl, CsCl, or ZnS type cubic structure. Predict the type of...
-
From Equation 8.4 and the ionic radii given in Figure 7.7, calculate the potential energy of the following pairs of ions. Assume that the ions are separated by a distance equal to the sum of their...
-
From the ionic radii given in Figure 7.7, calculate the potential energy of a Ca2+ and O2- ion pair that is just touching (the magnitude of the electronic charge is given on the back inside cover)....
-
2. Evaluate the following definite integrals (a) (2x - 6x) dx /6 (b) 16 cos 3t + 2 sin 3t dt x+1 2 da
-
Replace the two forces acting on the grinder by a resultant force and couple moment at point O. Express the results in Cartesian vectorform. F, = [101 - 15j - 40k} N 250 mm F; = |-151 20j 30OK} N...
-
In Exercises, determine whether the graph is that of a function. X
-
You are given two metal rods. One is a magnet, and the other is made of magnetic material but does not have the elementary magnets aligned. Using no other objects, how can you determine which is the...
-
Presented below is information related to Russell Corporation for the month of January 2012. InstructionsPrepare the necessary closingentries. Cost of goods sold Freight-out Salaries and wages...
-
n 1 1. lim tantan is equal to 004-1 1+r+r2 2. 3. If the least and the largest real values of a, for which the equation z + a |2-1+21=0(z=C and i=1) has a solution, are p and q respectively, then 4(p...
-
Metals with bcc structures, such as tungsten, are not close packed. Therefore, their densities would be greater if they were to change to a ccp structure (under pressure, for instance). What would...
-
Without doing a calculation, order the following gases according to increasing mass density: N 2 H 4 ; N 2 ; NH 3 . The temperature and pressure are the same for all three samples.
-
In Exercises 126, graph each inequality. x 1
-
What kinds of marketing problems might be addressed through: (a) exploratory; (b) descriptive; and (c) causal research projects?
-
What is a book, and what exactly is the core benefit that the reader derives from it? If its about engaging with the content, then the emergence of e-reading devices and digital books represents a...
-
What is environmental scanning, why is it important, and what are the potential problems of implementing it?
-
To what extent and why do you think that costs should influence pricing?
-
Explain the role that direct marketing can play in both creating and retaining customers.
-
What are the roles of IT, security, and general management with regard to physical security?
-
Use this circle graph to answer following Exercises. 1. What fraction of areas maintained by the National Park Service are designated as National Recreation Areas? 2. What fraction of areas...
-
Air bags are activated when a severe impact causes a steel ball to compress a spring and electrically ignite a detonator cap. This action causes sodium azide (NaN 3 ) to decompose explosively...
-
At elevated temperatures, sodium chlorate decomposes to produce sodium chloride and oxygen gas. A 0.8765-g sample of impure sodium chlorate was heated until the production of oxygen gas ceased. The...
-
Xenon and fluorine will react to form binary compounds when a mixture of these two gases is heated to 400 o C in a nickel reaction vessel. A 100.0-mL nickel container is filled with xenon and...
-
When the Centers for Medicare and Medicaid Services (CMS) makes a payment using the Prospective Payment System, what is the Medicare payment based on?
-
consent must be obtained from the patient, or from a person authorized to consent on the patient's behalf, before any medical procedure can be performed. Explain how consent differs among competent...
-
During the initial try-in of a three-unit fixed dental prosthesis (bridge), the prosthesis fits the prepared teeth perfectly and appears to be retained on the teeth so tenaciously that removal...

Study smarter with the SolutionInn App


