Evaluate the unknown quantity in each of the following cells: (a) Pt(s) H(g, 1.0 bar) |H* (pH
Question:
Evaluate the unknown quantity in each of the following cells: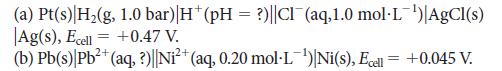
Transcribed Image Text:
(a) Pt(s) H₂(g, 1.0 bar) |H* (pH = ?)||Cl(aq,1.0 mol·L¹) AgCl(s) Ag(s), Ecell = +0.47 V. (b) Pb(s) Pb²+ (aq, ?)||Ni²+ (aq, 0.20 mol·L¹)|Ni(s), Ecell = +0.045 V.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To evaluate the unknown quantity in each cell we can use the Nernst equation 00592 Ecell Estandard c...View the full answer

Answered By

Rustia Melrod
I am a retired teacher with 6 years of experience teaching various science subjects to high school students and undergraduate students. This background enables me to be able to help tutor students who are struggling with the science of business component of their education. Teaching difficult subjects has definitely taught me patience. There is no greater joy for me than to patiently guide a student to the correct answer. When a student has that "aha!" moment, all my efforts are worth it.
The Common Core standards are a useful yardstick for measuring how well students are doing. My students consistently met or exceeded the Common Core standards for science. I believe in working with each student's individual learning styles to help them understand the material. If students were struggling with a concept, I would figure out a different way to teach or apply that concept. I was voted Teacher of the Year six times in my career. I also won an award for Innovative Teaching Style at the 2011 National Teaching Conference.
4.90+
4+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Evaluate the unknown quantity in each of the following cells: (a) Pt(s) |H(g, 1.0 bar) H*(pH = ?)||Cl(aq, 1.0 mol-L-) HgCl (s) Hg(1), Ecell = +0.33 V. (b) C(gr) C1(g, 1.0 bar) |Cl(aq, ?)||MnO4 (aq,...
-
The following eukaryotic structural gene contains two introns and three exons. exon 1 intron 1 exon 2 The table below shows four possible mRNA products of this gene. Use the labels to explain what...
-
If a nominal interest rate of 8% is compounded continuously, determine the unknown quantity in each of the following situations: a. What uniform EOY amount for 10 years is equivalent to $8,000 at EOY...
-
Horizon BP in Antelope Park, Alaska, has noticed that utility bills are substantially higher when the average monthly temperature is colder. The only thing in the shop that uses natural gas is the...
-
EOQ for manufacturer. Lakeland Company, which produces lawn mowers, purchases 18,000 units of a rotor blade part each year at a cost of $60 per unit Lakeland requires a 15% annual rate of return on...
-
The following jobs are waiting to be processed at the same machine center. Jobs are logged as they arrive: In what sequence would the jobs be ranked according to the following decision rules: (a)...
-
You are a salesperson working for American Airlines calling on the vice president of a large manufacturing company. Many of the companys people travel all over the world, and you would like them to...
-
A snowmobile manufacturer produces three models, the XJ6, the XJ7, and the XJ8. In any given production-planning week, the company has 40 hours available in its final testing bay. Each XJ6 requires 1...
-
1.) Identify the price, place and promotion strategies of Apple Pay? How is Apple Pay being promoted in the US. - How, When, Where, by what means. 2.) How does their marketing strategy reflect the...
-
Estimate the pH of (a) 0.0153 m NaHCO 3 (aq); (b) 0.110 m KHCO 3 (aq).
-
What is the concentration of hydronium ions in (a) A solution that is 0.12 m HBrO(aq) and 0.160 m NaBrO(aq); (b) A solution that is 0.250 m CH 3 NH 2 (aq) and 0.150 m CH 3 NH 3 Cl(aq); (c) A solution...
-
One criticism of these experiments might be that the presence of more than one sample of bacteria in each Petri dish might have had an effect on the results. Which of the following changes in...
-
Draw a sawtooth diagram with the above information. Noted that the retention money should be $35000x5% at the beginning and reduced to $35000x2.5% at the contract completion. The profit is for each...
-
The equations for IDFT (Inverse Discrete Fourier Transform-x[n]) and DFT (Discrete Fourier Transform-X[*]) are defined as It is given that x[n] =x[k]e * k=0~(N-1) X[k]=x[n]e -0 n=0~(N-1) -1/2k [x[n]...
-
IssueTracking System (ITS) Description An Issue Tracking System (ITS) isa computer software application that manages and maintains lists ofissues, as needed by departments of an organization to help...
-
M11 L1 - Activity 1: Static Electricity Investigation Lab Static Electricity Investigation Part 1: Electric Charges Lab Directions: Complete the Static Electricity Investigation Part 1: Electric...
-
The Coffee Table Company Ltd was established on 31st December 2005 to manufacture coffee tables for sale to the furniture retail trade. The owners invested K100 million as share capital. A long term...
-
The following are the income statements of two firms in the same industry. Required: a. Compute the contribution margin ratio, profit margin ratio, and sales per $ in fixed cost for the two firms. b....
-
An auto-parts manufacturer is considering establishing an engineering computing center. This center will be equipped with three engineering workstations each of which would cost $25,000 and have a...
-
Fill in the missing reagents: .N -NH2 -NH2
-
In this chapter, we explained why pyrrole is such a weak base, but we did not discuss the acidity of pyrrole. In fact, pyrrole is 20 orders of magnitude more acidic than most simple amines. Draw the...
-
Rimantadine is an antiviral drug used to treat people infected with life-threatening influenza viruses. Identify the starting ketone that would be necessary in order to prepare rimantadine via a...
-
What are the Strengths/Weaknesses of the organization's key financial Organization: The Make-A-Wish Foundation Link: https://wish.org/ Links to financial reports https://wish.org/managing-our-funds...
-
1. A company has three bonds they've issuedone's with coupon rates of 4.5%, 4.75%, and 5.2%. Together, the bonds are listed on their SFP at $300 million. The three have been priced in the market to...
-
For the fiscal year ending December 31, 2022, a companys total debt was $54.51 billion and total shareholder's equity was $81.45 billion. Based on these figures, the debt-to-equity ratio is .67. What...

Study smarter with the SolutionInn App


