Question: Name each of the following molecules. (a) (b) H CH3CHCH CH3 C=C CH3 H
Name each of the following molecules.
(a)
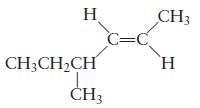
(b)
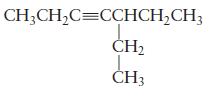
H CH3CHCH CH3 C=C CH3 H
Step by Step Solution
3.44 Rating (151 Votes )
There are 3 Steps involved in it
a The longest chain which contains six carbon atoms is number... View full answer

Get step-by-step solutions from verified subject matter experts


