Predict the major products of each of the following reactions and then balance the equations: (a) TiCl4(s)
Question:
Predict the major products of each of the following reactions and then balance the equations: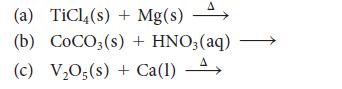
Transcribed Image Text:
(a) TiCl4(s) + Mg(s) A (b) COCO3(s) + HNO3 (aq) (c) V₂05(s) + Ca(1) A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a TiCl g 2 Mg1 Tis ...View the full answer

Answered By

Joram mutua
I am that writer who gives his best for my student/client. Anything i do, i give my best. I have tutored for the last five years and non of my student has ever failed, they all come back thanking me for the best grades. I have a degree in economics, but i have written academic papers for various disciplines due to top-notch research Skills.In additional, I am a professional copywriter and proofreader.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Predict the major products of the following reactions. Include stereochemistry where applicable. (a) 1-methylcycloheptene + BH3 THF, then H2O, OH- (b) Trans-4,4dimethylpent-2ene + BH THF, then H2O2,...
-
Predict the major products of the following reactions: (a) (c) (e) Product of (c) + H2O Product of (c) + H2 (1 molar equivalent) (g) Product of (f) HCI Styrene EtONa HA, heat OH peroxides HA heat Pt...
-
Predict the major products of the following reactions. (a) (E)-3-methyloct-3-ene + ozone, (CH3)2S (b) (Z) -3- methyloct-3ene + warm, concentrated KMnO4 (d) I-ethylcycloheptene +ozone, then (CH3)2S...
-
. What is the output of the following program? Write your answer in the box below. #include using namespace std; int main() { int a=4; int b=6; int i; int j; } int** k; i = &a; j = &b; *j = 8; if...
-
Provide a scenario in which a process executing the V procedure in Figure will detect when s. value is less than or equal to 0 and s. hold is TRUE. struct semaphore ( int value = ; boolean mutex =...
-
Shields Manufacturing produces containers for nurseries and landscaping businesses. The company competes based on its low-cost, high-quality products. As the company's expenses have risen, management...
-
For each of the following situations, calculate the degrees of freedom \((d f)\), identify the critical values (assume \(\mathrm{a}=.05\) [two-tailed]), calculate the standard error of the mean \(s\)...
-
Air travel on Mountain Airlines for the past 18 weeks was: a. Explain why an averaging technique would not be appropriate for forecasting. b. Use an appropriate technique to develop a forecast for...
-
Each question below is 1 point each, for a total of 3 extra credit points. There is no time limit on this. 1. What are free cash flows? 2. Explain the difference between a company's operating cash...
-
Identify the number of valence electrons (including d-electrons) present in each of the following metal ions: (a) Ti 2+ ; (b) Tc 2+ ; (c) Ir + ; (d) Ag + ; (e) Y 3 + ; (f) Zn 2 + .
-
Which members of the d-block, those at the left or at the right of the block, are likely to have the more strongly negative standard potentials? Explain your prediction.
-
Multiple Choice Questions: 1. Which of the following is not a possible information system security deliverable? a. Software. b. Hardware. c. Personnel. d. None of the above. e. All of the above. 2....
-
BQW's United States (US) regional division reported $40,000 of revenue, $13,000 of variable costs and $10,000 of traceable fixed costs. Common fixed costs amounted to $2,500. What was the segment...
-
A carpenter holds a 14-lb 2-in. by 4-in. board as shown. If he exerts vertical forces on the board, determine the forces at A and B (use positive if the force is up and negative if down). A Answers:...
-
Compare the volume required for a completely mixed flow reactor to that of a plug flow reactor to achieve a 104 reduction in the coliform count of a treated wastewater effluent. As per Chick's Model,...
-
Gamma acquired 60% of Delta's shares on January 1, 2024 paying $500,000 in cash. Delta's balance sheet as of January 1, 2024 and the fair value of Delta's assets and liabilities were as follows:...
-
ps://mybscbryantstratton.edu/webapps/assessment/take/launch.jsp?course_assessment_id=_361377_18course_id=_93... A Question Completion Status: QUESTION 10 8.5 points Compose a statement that correctly...
-
Explain the terms nouvelle cuisine and fusion cuisine.
-
On average there are four traffic accidents in a city during one hour of rush-hour traffic. Use the Poisson distribution to calculate the probability that in one such hour there arc (a) No accidents...
-
What is meant by the weight of a configuration?
-
How does one calculate the number of microstates associated with a given configuration?
-
Describe what is meant by the phrase the dominant configuration.
-
Vice President for Sales and Marketing at Waterways Corporation is planning for production needs to meet sales demand in the coming year. He is also trying to determine how the company's profits...
-
How Complex Systems Fail Did you look at the video presentation by Steven Spear? What is a complex system? Are large organizations complex systems? What were the key lessons learnt by the author in...
-
In what ways can parametric design methodologies redefine spatial configurations and architectural aesthetics ?

Study smarter with the SolutionInn App


