Predict the potential of each of the following cells: (a) Pt(s) H(g, 1.0 bar) |HCl(aq, 0.075 mol-L)||HCl(aq,
Question:
Predict the potential of each of the following cells: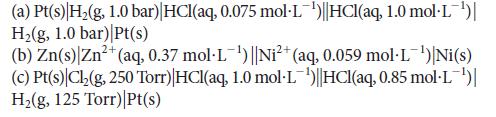
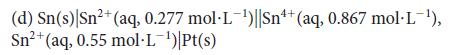
Transcribed Image Text:
(a) Pt(s) H₂(g, 1.0 bar) |HCl(aq, 0.075 mol-L¯¹)||HCl(aq, 1.0 mol-L¯¹)| H₂(g, 1.0 bar) |Pt(s) (b) Zn(s) Zn²+ (aq, 0.37 mol·L¹) ||Ni²+ (aq, 0.059 mol·L¯¹)|Ni(s) (c) Pt(s) Ch₂2(g, 250 Torr) |HCl(aq, 1.0 mol·L¹)||HCl(aq, 0.85 mol·L¯¹)| H₂(g, 125 Torr) |Pt(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
a 0067 ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Water potential in potato cells was determined in the following manner. The initial masses of six groups of potato cores were measured. The potato cores were placed in sucrose solution of various...
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
The owners want to do some analysis to see how their sales are doing. Management wants to see how the company did last year and make some decisions about what products to keep along with staffing for...
-
3. In this problem the bond sells for a premium, which indicates that interest rates have declined. The current yield is $70/$1,222 = 5.73%. To confirm that interest rates have declined, determine...
-
Reworked units, casts of rework. White Goods assembles washing machines at its Auburn plant. In February 2009, 60 tumbler units that cost $44 each (from a new supplier who subsequently went bankrupt)...
-
Tim Smunt has been asked to evaluate two machines. After some investigation, he determines that they have the costs shown in the following table. He is told to assume that: (a) The life of each...
-
Who can conduct cost audit under the Companies Act, 2013?
-
An insurance company has high levels of absenteeism among the office staff. The head of office administration argues that employees are misusing the companys sick leave benefits. However, some of the...
-
Warnerwoods Company uses a perpetual inventory system. It entered into the following purchases and sales transactions for March. Date Units Acquired at Cost: March 1 Activities Beginning inventory...
-
Calculate the volume of 0.150 m HCl(aq) required to neutralize (a) one-half and (b) all the hydroxide ions in 25.0 mL of 0.110 m NaOH(aq). (c) What is the molar concentration of Na + ions at the...
-
(a) Give the Ka value of each of the following acids: (i) Phosphoric acid, H 3 PO 4 , pK a = 2.12; (ii) Phosphorous acid, H 3 PO 3 ,pK a = 2.00; (iii) Selenous acid, H 2 SeO 3 , pK a = 2.46; (iv)...
-
Determine whether the statement is true or false. If it is true, explain why. If it is false, explain why or give an example that disproves the statement. If F and G are vector fields and divF =...
-
For Experiment 3 in Chem 255 Lab, students need to extract the fat trimyristin from the common cooking spice nutmeg before it can be converted to myristic acid. A student was able to extract 2.65 g...
-
3. Scenario/Introduction/Background Information: 1909 Drink Sdn Bhd is a newly established company which produces bottled soya bean drink. The company was incorporated on 1" January 2021 and...
-
You are submitting an application to a professional conference on data-driven decision-making. The application has two parts: Providing information to the conference organizers. Creating a...
-
Bill Bostic, Sue Chung, and Sam De Felice were assigned ihe task of explaining the elements of financial management to PNC's board of directors, ard the rext session will deal with capital structure....
-
This assignment asks you to use the techniques that you have learned in this course to evaluate a potential investment opportunity. Specifically, it asks you to use the data provided with this...
-
Ricardo Windows specializes in making custom windows for high-end homes. Although the demand for custom windows fluctuates from year to year, the owner, Javier Ricardo, believes that it is in his...
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
How did Planck conclude that the discrepancy between experiments and classical theory for blackbody radiation was at high and not low frequencies?
-
Write down formulas relating the wave number with the frequency, wavelength, and energy of a photon.
-
Plancks explanation of blackbody radiation was met by skepticism by his colleagues because Equation (12.4) seemed like a mathematical trick rather than being based on a microscopic model. Justify...
-
Selected data for Adams Company for Year 3 follows: Earnings (net income) $ 184,000 Preferred stock (13,000 shares at $50 par, 6%) $ 650,000 Common stock (44,000 shares no par, market value $47)...
-
How do regulatory frameworks and compliance standards, such as the Payment Card Industry Data Security Standard (PCI DSS) and the European Unions Revised Payment Services Directive (PSD2), influence...
-
Spiderman Corp. has two departments. The following are the data of the two departments. Quantity Department 1 Department 2 Beginning Work in Process 32,500 22,500 Incomplete Material 20% 30%...

Study smarter with the SolutionInn App


