The following are representations of acidbase reactions: + 11 + +
Question:
The following are representations of acid–base reactions: 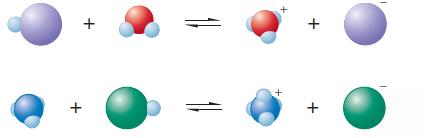
Transcribed Image Text:
+ 11 呢 + +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
HCl H2O H3...View the full answer

Answered By

Godswill Okorie
M.Sc chemistry specialization in organic chemistry, B.ed .I am having industry experience of seven years working with Ranbaxy nd Shimadzu analytical India by working as an application chemistry.I am having good practical experience on chromatography techniques,which later helped me in my teaching.I worked as PGT chemistry teacher with KV and APS.
As a teacher I was able to achieve good results with my students.I used to take 11th and 12th chemistry and science to classes 7th ,8th and ninth. While teaching I used to guide students for various carrier opportunities.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following are representations of acid base reactions: a. Label each of the species in both equations as an acid or a base and explain. b. For those species that are acids, which labels apply:...
-
The following are representations of two forms of glucose. The six-membered ring is known to exist in a chair conformation in each form. Draw clear representations of the most stable conformation of...
-
The following are representations of two forms of glucose. The six-membered ring is known to exist in a chair conformation in each form. Draw clear representations of the most stable conformation of...
-
M1 is a way to measure... a) the level of bank reserves b) a country's money supply c) the level of savings in a country d) a country's economic potential
-
Suppose that X and Y are independent random variables, that X has the uniform distribution on the integers 1, 2, 3, 4, 5 (discrete), and that Y has the uniform distribution on the interval [0, 5]...
-
A turbine blade made of a metal alloy (k=17 W/mK) has a length of 5.3 cm, a perimeter of 11 cm, and a cross-sectional area of 5.13 cm 2 . The turbine blade is exposed to hot gas from the combustion...
-
How are COTS products procured?
-
Neil Corporation has three projects under consideration. The cash flows for each project are shown in the following table. The firm has a 16% cost of capital. a. Calculate each project's payback...
-
What steps are needed to write equations for SOP and POS forms? CD AB 00 01 11 10 AB 00 1 1 1 0 00 00 H 01 0 0 1 1 01 0 CD 00 01 11 10 1 1 1 0 1 1 0 11 1 0 1 1 11 1 0 1 1 10 0 1 1 0 10 0 1 1 0
-
Pedro and Lucy started to create a budget (based on last years income and expense statement) but got stuck. They know that you have learned how to create budgets and are asking for your help. They...
-
You have two solutions of the salts NaX(aq) and NaY(aq) at equal concentrations. What would you need to know to determine which solution has the higher pH? Explain how you would decide (perhaps even...
-
Acids and bases can be thought of as chemical opposites (acids are proton donors, and bases are proton acceptors). Therefore, one might think that K a = 1/K b . Why isnt this the case? What is the...
-
Why does a need typically have to be recognized before it will motivate?
-
Compute the limit of the convergent sequences in Problems 19-26. \(\left\{\frac{8 n^{3}-6 n^{2}+85}{2 n^{3}-5 n+170}ight\}\)
-
Find each limit in Problems 11-18, if it exists. \(\lim _{n ightarrow \infty} \frac{n}{5,000-n}\)
-
Consider the sequence \(6,6.6,6.66,6.666, \cdots\). What do you think is the appropriate limit of this sequence?
-
The SAT scores of entering first-year college students are shown in Figure 18.21. In Problems 13-18, find the average yearly rate of change of the scores for the requested periods. Figure 18. 21 2008...
-
Compute the limit of the convergent sequences in Problems 19-26. \(\left\{\frac{8 n^{2}+800 n+5,000}{2 n^{2}-1,000 n+2}ight\}\)
-
Suppose you have a $200,000 home loan with an annual interest rate of 6.5%, compounded monthly. a. If you pay $1200 per month, what balance remains after 20 years? b. If you pay $1400 per month, what...
-
The figure shows a bolted lap joint that uses SAE grade 8 bolts. The members are made of cold-drawn AISI 1040 steel. Find the safe tensile shear load F that can be applied to this connection if the...
-
A mixture of chromium and zinc weighing 0.362 g was reacted with an excess of hydrochloric acid. After all the metals in the mixture reacted, 225 mL of dry hydrogen gas was collected at 27 o C and...
-
You have a sealed, flexible balloon filled with argon gas. The atmospheric pressure is 1.00 atm and the temperature is 25oC. The air has a mole fraction of nitrogen of 0.79, the rest being oxygen. a....
-
Derive a linear relationship between gas density and tem-perature, and use it to estimate the value of absolute zero temperature (in o C to the nearest 0.1 o C) from an air sample whose density is...
-
7. Ionizing radiation involves all of the following except a. UV light b. Gamma rays c. Electron beams d. X-rays 8. The term zone of inhibition is used in which of the following procedures? a....
-
4. Which of the following methods is the simplest heat-related method to sterilize metal? a. Incineration b. Autoclaving c. Direct flaming d. Indirect heating 5. Boiling items in water for minutes...
-
Summary: The following data is provided for a product to cost-volume-profit analysis. Original Data Sales price per unit $50.00 Variable costs per unit $23.00 Fixed Costs $5,500 Monthly Volume 250...

Study smarter with the SolutionInn App


