The following Lewis structure was drawn for a Period 3 element. Identify the element. CI :0: E-
Question:
The following Lewis structure was drawn for a Period 3 element. Identify the element.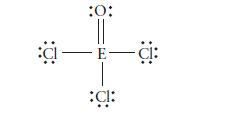
Transcribed Image Text:
CI :0: E- :15: - CI:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
E i...View the full answer

Answered By

Raunak Agarwal
Teaching is my hobby and now my profession. I teach students of CA and CFA(USA) in batches of 100 students and have a 5 year experience.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The following Lewis structure was drawn for a Period 4 element. Identify the element. :0: E=0 :CI:
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
A second Lewis structure can be drawn for one of the nucleophiles in Problem 36. (a) Identify it and draw its alternate structure (which is simply a second resonance form), (b) Is there a second...
-
Many auditors consider the substantiation of the figure for inventory to be a more difficult and challenging task than the verification of most other items on the balance sheet. List several specific...
-
Prepare the journal entries that the lessee should make to record the following transactions. 1. The lessee makes a lease payment of $80,000 to the less or in an operating lease transaction. 2....
-
Decide whether integration by parts or a substitution should be used to compute the indefinite integral. If substitution, indicate the substitution to be made. If by parts, indicate the functions f...
-
Daniel lives in a rural town located in the Commonwealth of Massachusetts on 200 acres of land that includes a house, a greenhouse, and a large garden. The greenhouse is located about 15 feet from...
-
Rainger Company is considering replacement of some of its manufacturing equipment. Information regarding the existing equipment and the potential replacement equipment follows. Required Based on this...
-
1. Define ADT (Abstract Data Type)? 2. Mention the features of ADT.? 3. Define List ADT? 4. What are the ways of implementing linked list? 5. What are the types of linked lists?
-
Identify the hybrid orbitals used by the atom in boldface red type in each of the following species: (a) BF 3 ; (b) AsF 3 ; (c) BrF 3 ; (d) SeF 3 + .
-
State the hybridization of the atom in boldface red type in each of the following molecules and ions: (a) SF 6 ; (b) ClO 3 ; (c) NO 3 ; (d) OCCl2 .
-
Rewrite each expression using the properties of exponents so that the variable appears only once. a. (4x2)(x) b. 4x2 / 8x3 c. (x3)5
-
John Jefferson dies on November 10, 2019. John is on the cash basis of accounting for tax purposes. On November 29, 2019, a $4,000 paycheck covering the period November 1 November 8, 2019, is mailed...
-
Mary Beth is a CPA, devoting 3,000 hours per year to her practice. She also owns an office building in which she rents out space to tenants. She devotes none of her time to the management of the...
-
Richard Johnson held $50,000 of U.S. government savings bonds on the date of his death, December 11, 2019. The bonds mature in 2020. Unreported interest income on these bonds at the date of Richard's...
-
The trust instrument requires the trustee to distribute $40,000 annually to Carl Smith, the grantor's son. Any residual income may be distributed or accumulated for Bob Jones, Sam Smith, Earl Litt,...
-
Hershel Barker was the president and majority shareholder in Bulldog Inc. He was a cash-basis taxpayer who reported his income on a calendar-year basis. On March 1, 2019, Hershel was killed in a...
-
The bulk modulus of water is B = 2.2 109 N/m2. What change in pressure P (in atmospheres) is required to keep water from expanding when it is heated from 15 to 25C?
-
Represent each of the following combination of units in the correct SI form using an appropriate prefix: (a) m/ms, (b) k m, (c) k s /mg, and (d) k m N.
-
A solution contains 1.0 3 1026 M HOCl and an unknown concentration of KOCl. If the pH of the solution is 7.20, calculate the KOCl concentration.
-
A solution contains 1.0 3 1026 M HOCl and an unknown concentration of KOCl. If the pH of the solution is 7.20, calculate the KOCl concentration.
-
An aqueous solution contains dissolved C 6 H 5 NH 3 Cl and C 6 H 5 NH 2 . The concentration of C 6 H 5 NH 2 is 0.50 M and pH is 4.20. a. Calculate the concentration of C 6 H 5 NH 3 + in this buffered...
-
P.3.3 Show that the one-dimensional Navier-Stokes equation without pressure gradient (known as the 'viscous' Burger's equation) au +u=- at ax 2x2 is parabolic in x, t. Hint: Write the equation as a...
-
d. Blood is mostly water, but has a lower surface tension (~55 mN/m), why? (what contents in blood attribute to the surface tension reduction? Why?
-
Average Penalty Minutes in the NHL An interval estimate was constructed for mean penalty minutes given to NHL players in a season using data from players on the Ottawa Senators as our sample. Some...

Study smarter with the SolutionInn App


