The hydrolysis of an organic nitrile, a compound containing aCN group, in basic solution, is proposed to
Question:
The hydrolysis of an organic nitrile, a compound containing a—C≡N group, in basic solution, is proposed to proceed by the following mechanism. Write a complete balanced equation for the overall reaction, list any intermediates, and identify the catalyst in this reaction.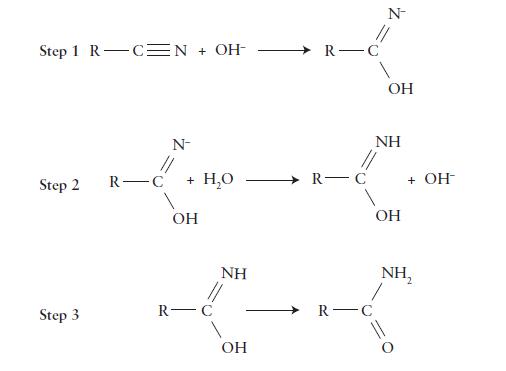
Transcribed Image Text:
Step 1 R C N + OH- Step 2 Step 3 R-C N- + H₂O OH R- NH OH R-C R-C R-C N OH NH OH + OH- NH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
RCN HO RCN...View the full answer

Answered By

Sultan Ghulam Dastgir
The following are details of my Areas of Effectiveness English Language Proficiency, Organization Behavior , consumer Behavior and Marketing, Communication, Applied Statistics, Research Methods , Cognitive & Affective Processes, Cognitive & Affective Processes, Data Analysis in Research, Human Resources Management ,Research Project,
Social Psychology, Personality Psychology, Introduction to Applied Areas of Psychology,
Behavioral Neurosdence , Historical and Contemporary Issues in Psychology, Measurement in Psychology, experimental Psychology,
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Consider the following mechanism for the hydrolysis of ethyl acetate. Write a complete balanced equation for the overall reaction, list any intermediates, and identify the catalyst in this reaction....
-
Sondra is the leader of a nonprofit organization with about 40 full- and part-time employees. She is proud of her organization and its very dedicated staff. Sondra has been with the organization for...
-
The following mechanism has been proposed for the gas-phase reaction of H2 with ICl: H2(g) + ICl(g) HI(g) + HCl(g) HI(g) + ICl(g) I2(g) + HCl(g) (a) Write the balanced equation for the overall...
-
No Opportunity Costs The Van Division of MotoCar Corporation has offered to purchase 180,000 wheels from the Wheel Division for $41 per wheel. At a normal volume of 500,000 wheels per year,...
-
Figure is the simplified process state diagram with running, ready, and blocked states. Modify the diagram so that it includes new states to represent when a process is swapped out. Show the...
-
Company reports the following financial information before adjustments. Instructions Prepare the journal entry to record Bad Debt Expense assuming Rodriguez Company estimates bad debts at (a) 3% of...
-
Search the literature or web and discuss briefly the principles behind the following flowmeasurement devices: a Pitot tube; a hot-wire anemometer; a laser-Doppler velocity meter; and a...
-
For each independent case, fill in the missingfigures. Case A Case B Case C Case D Units produced Standard hours per unit Standard hours Standard rate per hour Actual hours worked Actual labor cost...
-
What is the error to this Matlab code for part 3b? Please do parts 3b - 3e Image transcription text WC=1; [-,idx] = min (abs (wout - wc) ) ; A = mag (idx) ; - theta = phaseDeg (idx) ; DO YOUIA WN t...
-
The following mechanism has been proposed for the gasphase reaction between HBr and NO 2 : (a) Write the overall reaction. (b) Write the rate law for each step and indicate its molecularity. (c) What...
-
The rate constant of the reaction between CO 2 and OH in aqueous solution to give the HCO 3 ion is 1.5 * 10 10 L mol 1 s 1 at 25C. Determine the rate constant at human body temperature (37C),...
-
Carfly Barbers decides to lease another barbershop for a 10- year period. The barbershop has a cash price of $ 200,000. If Carfly borrowed money to purchase the shop, it would have had to pay 12%...
-
Discuss the role of the government sponsored-enterprises (GSEs) in the funding and issuance of mortgage loans.
-
Permission granted to buy conventional mortgages, which were not government guaranteed, opened doors to the formation of new types of mortgage companies that expanded into __________ subprime loans.
-
The 80/20 loan is a mortgage loan whereby the borrower takes out a loan for 80 percent of the purchase price and a second loan for 20 percent of the purchase price, leaving the borrower with no...
-
Before the passage of the Secondary Mortgage Market Enhancement Act of 1984, there were regulations that prohibited __________ investors from investing in mortgage-backed securities that were not...
-
Tranching is a process of carving up or separating a pool of mortgages into different classes of securities that absorb different __________ losses.
-
A corporation has two departments that produce two separate product lines. The company has been implementing total quality management over the past year. Revenue and costs of quality for that year...
-
Assume a simple Keynesian depression economy with a multiplier of 4 and an initial equilibrium income of $3,000. Saving and investment equal $400, and assume full employment income is $4,000. a. What...
-
For each of the following substrates, determine whether an SN1 process will require a proton transfer at the beginning of the mechanism: (a) (b) (c) (d) (e) (f) -OH
-
Will an S N 1 process involving each of the following nucleophiles require a proton transfer at the end of the mechanism? (a) NaSH (b) H 2 S (c) H 2 O (d) EtOH (e) NaCN (f) NaCl (g) NaNH 2 (h) NH 3...
-
For each of the following substrates, determine whether an S N 1 process will involve a carbocation rearrangement or not: (a) (b) (c) (d) (e) (f)
-
What components of company annual reports provides the numbers that investors use to determine how well tge company performed financially during a given period?
-
Paul works in an organizational structure where the lines of communication are very clear; however, managers often feel isolated and lack the resources needed to achieve company goals. Paul most...
-
Components within your server room are failing at a rapid pace. You discover that the humidity in the server room is at 6 0 % and the temperature is at 8 0 degrees. What should you do to help reduce...

Study smarter with the SolutionInn App


