The mechanism proposed for the oxidation of iodide ion by the hypochlorite ion in aqueous solution is
Question:
The mechanism proposed for the oxidation of iodide ion by the hypochlorite ion in aqueous solution is as follows: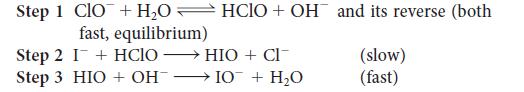
Write the rate law for the formation of HIO implied by this mechanism.
Transcribed Image Text:
Step 1 ClO + H₂0HCIO + OH and its reverse (both fast, equilibrium) Step 2 I + HClO →→ HIO + Cl¯ Step 3 HIO + OH- → 10 + H₂O (slow) (fast)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The rate law for the formation of HIO implied by the mechanism is rate kIHClO Herek is the rate cons...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The oxidation of iodide ion by the hypochlorite ion in the presence of hydroxide ions was studied at 25C, and the following initial rates data (Y. Chia and R. E. Connick, Journal of Physical...
-
The iodide ion reacts with hypochlorite ion (the active ingredient in chlorine bleaches) in the following way: OCI + IË -- OIË + CIË. This rapid reaction gives the following rate data:...
-
The oxidation of iodide ion by arsenic acid, H3AsO4, is described by the balanced equation (a) if ? ?[I] / ?t = 4.8 ? 104 M/s, what is the value of ? [13] / ?f during the same time interval? (b) What...
-
Shreya commutes in a CNG fitted van to school every day along with many other students. She told the van driver to get the CNG connection certified and timely checked it for any leakage or loose...
-
Suppose hardware was designed to incorporate three reference bits rather than the single bit used in the LRU implementation described in Section 12.3. Explain how a better approximation to LRU could...
-
Soundgarden Company sold 200 color laser copiers in 2014 for $4,000 apiece, together with a one-year warranty. Maintenance on each copier during the warranty period averages $330. Instructions (a)...
-
Consider a $2^{2}$ factorial experiment with factors A and B. Show that $I N T(A, B)=I N T(B, A)$. That is, the interaction is symmetric in $\mathrm{B}$ and A.
-
Steve and Sue are married with three dependent children. Their 2016 joint income tax return shows $389,000 of AGI and $60,000 of itemized deductions made up of $30,000 of state income taxes and...
-
Marketing Analytics 6.1 The Expanding Uses of Data Mining and Analytics in the Restaurant Industry** When Damian Mogavero, a restaurant group CFO, realized the vast need for improved data analytics...
-
Rate laws for gas-phase reactions can also be expressed in terms of partial pressures, for instance, as Rate 5 k r P J for a firstorder reaction of a gas J. What are the units for the rate constants...
-
Decide which of the following statements about catalysts are true. If the statement is false, explain why. (a) A heterogeneous catalyst works by binding one or more of the molecules undergoing...
-
Evaluate the following integrals. Je e* sin 4x dx
-
Describe and compare Lockheeds capitalized development costs and Livents capitalized preproduction costs.
-
How did the development costs at Lockheed and the preproduction costs at Livent differ from preproduction costs related to long-term supply arrangements?
-
The SECs enforcement action against Livent alleged that senior company officials had a special computer program designed that would enable accounting personnel to __________ the accounting system...
-
Explain how Livents improper deferral of expenses and Rite Aids improper recognition of vendor rebates shifted the recognition of expenses from one period to a later period.
-
Explain why Rite Aids failure to write down its ending inventory amountto agree with values determined by physical inventory countscaused it to overstate its income.
-
Park Electronics Company is known for its high-quality products and on time deliveries. Six months ago, it installed a computer-integrated manufacturing system in its Sensitive Components Department....
-
You purchase a bond with a coupon rate of 6.7 percent, a par value $1,000, and a clean price of $905. Assume a par value of $1,000. If the next semiannual coupon payment is due in two months, what is...
-
Draw all constitutional isomers that have molecular formula C 4 H 10 O?
-
Draw a Lewis dot structure for each of the following atoms: (a) Carbon (b) Oxygen (c) Fluorine (d) Hydrogen (e) Bromine (f) Sulfur (g) Chlorine (h) Iodine
-
Rank the indicated bonds in terms of increasing bond length: . 1 =C .
-
Description/ ($) Variable Fixed Direct Cost Cost Product Cost Material Labour S Direct Mfg. Sell. & Admin. y Opportunit Sunk Cost Overhea d Cost
-
Design for childcare centres a quality of : - Art Corner -construction corner And what Theories/ policies?
-
Some people have suggested that everyone between the ages of 18 and 21 should be required to perform one year of community or government service, such as in the Peace Corps, Environmental Conservancy...

Study smarter with the SolutionInn App


