Which of the following reactions can be classified as reactions between Brnsted acids and bases? For those
Question:
Which of the following reactions can be classified as reactions between Brønsted acids and bases? For those that can be so classified, identify the acid and the base.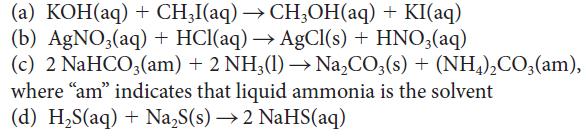
Transcribed Image Text:
(a) KOH(aq) + CH3I(aq) → CH3OH(aq) + KI(aq) (b) AgNO3(aq) + HCl(aq) → AgCl(s) + HNO3(aq) (c) 2 NaHCO3(am) + 2 NH3(1)→→ Na₂CO3(s) + (NH4)₂CO3(am), where "am" indicates that liquid ammonia is the solvent (d) H,S(aq) + Na,S(s) → 2 NaHS(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a KOHaq CH3Iaq CH3OHaq KIaq This reaction involves the transfer of a proton H from CH3I t...View the full answer

Answered By

Patrick Busaka
I am a result oriented and motivated person with passion for challenges because they provide me an opportunity to grow professionally.
5.00+
38+ Reviews
58+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
Proteins are synthesized with a particular amino acid sequence through the translation of information encoded in messenger RNA by an RNAprotein complex called a ribosome. Amino acids are specified by...
-
1. Which one of the following is Not an assumption of the binomial distribution? The probability of the success, p, is constant. The probability of the success is equal to 0.5 in all trials. There...
-
Consider X[k] be the N-point DFT of an N-point sequence x[n]. (20 %) x [n] = {1, -2, 1, 3} x [n] = {0, 2, -1,0,0,4} (a) Determine the linear convolution x [n] * x [n] (5%) (b) Determine the...
-
Allocation of common costs. Mike and Ed are students at Berkeley College. They share an apartment that is owned by Ed. Ed is considering subscribing to an Internet provider that has the following...
-
Sales of vegetable dehydrators at Bud Baniss discount department store in St. Louis over the past year are shown below. Management prepared a forecast using a combination of exponential smoothing and...
-
What are the most powerful social media networks that researchers could obtain data from about customer feedback and loyalty and why?
-
1. Calculate the incremental, or marginal, cost per chair to LP of accepting the order from Southeast. 2. What assumptions did you make in calculating the incremental cost in Question 1? What...
-
My question is: The article uses the term 'self-efficacy' several times. Briefly describe what you understand by self-efficacy in the context of the paper and whether you think it has been clearly...
-
Use the data in Appendix 2B to calculate E(Ti 3+ /Ti). 2B STANDARD POTENTIALS AT 25 C Potentials in Electrochemical Order Reduction half-reaction Strongly oxidizing H4XeO6 + 2 H+2 e XeO3 + 3 HO F +2e...
-
(a) Write balanced half-reactions for the redox reaction between sodium perchlorate and copper (I) nitrate in an acidic solution. (b) Write the balanced equation for the cell reaction and devise a...
-
PAR AND NO-PAR, COMMON AND PREFERRED STOCK Valdez Company had the following stock transactions during the year: (a) Issued 24,000 shares of $1 par common stock for $26,000 cash. (b) Issued 20,000...
-
1. The converter above is used to supply three identical, 50-Hz AC loads, represented by y, from a DC source (with Vin = 100V) using three half-bridge circuits. Assume the load impedance y has the...
-
Would your answer to Question 1.2 change if you were to finance the asking price of each building with a 50% loan-to-value mortgage? The loan bears interest at a rate of 4% and can be rolled over...
-
The Australian dollar is regarded as a commodity currency. Assuming that demand for iron ore is not affected by changes in commodity prices, this implies that an increase in the commodity price...
-
GBI management is seriously considering marketing a ruggedized version of their Touring Bike to "City Bike" bicycle rental programs. As a pilot, they have agreed to sell 30 City Bikes to a...
-
"Home" has 100 units of labor that are specific to the production of R&D, and 25 units of labor that are specific to the production of components; there are 100 units of capital that can be allocated...
-
Damon and Company uses the weighted-average method of process costing. On January 1, the units in work-in-process (WIP) inventory were 100% complete with respect to materials and 60% complete with...
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
An ideal solution is formed by mixing liquids A and B at 298 K. The vapor pressure of pure A is 151 Torr and that of pure B is 84.3 Torr. If the mole fraction of A in the vapor is 0.610, what is the...
-
A solution is prepared by dissolving 45.2 g of a nonvolatile solute in 119 g of water. The vapor pressure above the solution is 22.51 Torr and the vapor pressure of pure water is 23.76 Torr at this...
-
A sample of glucose (C 6 H 12 O 6 ) of mass 13.2 g is placed in a test tube of radius 1.25 cm. The bottom of the test tube is a membrane that is semi-permeable to water. The tube is partially...
-
Can the conceptual framework of bankruptcy be reconceptualized through interdisciplinary lenses, incorporating insights from behavioral economics, game theory, and institutional economics, to...
-
Martha is considering the purchase of a new oven for her bakery. Given the following information, use discount factors of 0.909 in year 1, 0.826 in year 2, and 0.751 in year 3, calculate the total...
-
Why is registration of a partnership is important?

Study smarter with the SolutionInn App


