A buffer is 0.100 M in NH 4 Cl and 0.100 M in NH 3 . When
Question:
A buffer is 0.100 M in NH4Cl and 0.100 M in NH3. When a small amount of hydrobromic acid is added to this buffer, which buffer component neutralizes the added acid?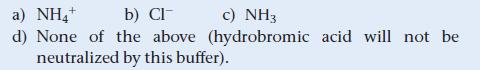
Transcribed Image Text:
a) NH4+ b) Cl c) NH3 d) None of the above (hydrobromic acid will not be neutralized by this buffer).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
c...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When a small amount of iodine is added to a mixture of chlorine and methane, it prevents chlorination from occurring. Therefore, iodine is a free-radical inhibitor for this reaction. Calculate Ho...
-
A solution is 0.0500 M in NH4Cl and 0.0300 M in NH3. Calculate its OH2 concentration and its pH (a) Neglecting activities. (b) Taking activities into account.
-
1) Which of the following solutions is a good buffer system? Which of the following solutions is a good buffer system? A solution that is 0.10 M HCN and 0.10 M LiCN A solution that is 0.10 M NaCl and...
-
In Problems 530, a. Classify the sequences as arithmetic, geometric, Fibonacci, or none of these. b. If arithmetic, give d; if geometric, give r; if Fibonacci, give the first two terms; and if none...
-
Many politicians, scientists, economists, and businesspeople have become concerned about the potential implications of global warming. The largest source of the emissions thought to contribute to...
-
Comparative balance sheet accounts of Sharpe Company are presented below. Additional data:1. Equipment that cost $10,000 and was 60% depreciated was sold in 2012.2. Cash dividends were declared and...
-
In 1988, the Upper Deck Company was a company with an idea for a better baseball card: one that had a hologram on it. By the 1990s, the firm was a major corporation worth at least a quarter of a...
-
Keep Cool Industries, Inc., manufactures fans for personal use. Department 70 is responsible for assembling the fan. Department 71 packages them for shipment. Keep Cool recently produced 10,000 fans...
-
1. Mr. i holds the following preferences over outcomes w, x, y, and z: xPw, xPy, zPx, yPz, wPy, and wPz. (a) Identify all five possible subsets of these outcomes that contain three or more outcomes....
-
Which solution is a buffer? (a) A solution that is 0.100 M in HNO 2 and 0.100 M in HCl (b) A solution that is 0.100 M in HNO 3 and 0.100 M in NaNO 3 (c) A solution that is 0.100 M in HNO 2 and 0.100...
-
Calculate the pH of a buffer solution that is 0.100 M in HC 2 H 3 O 2 and 0.100 M in NaC 2 H 3 O 2 .
-
The accurate control of a nuclear reactor is important for power system generators. Assuming the number of neutrons present is proportional to the power level, an ionization chamber is used to...
-
One reason the short-run aggregate-supply curve slopes upward is that a higher price level a. raises nominal wages if real wages are sticky. b. reduces nominal wages if real wages are sticky. c....
-
The benefit of diversification when constructing a portfolio is that it can eliminate a. adverse selection. b. risk aversion. c. firm-specific risk. d. market risk.
-
Desirae, an American college student, moves to Japan for the summer to teach English. Her salary is included a. only in U.S. GDP. b. only in Japans GDP. c. in both U.S. GDP and Japans GDP. d. in...
-
Jay arranges with First National Bank to make automatic monthly payments on his student loan. More than three days before a scheduled payment, Jay can stop the automatic payments by notifying the...
-
Which of the following would shift the aggregatedemand curve to the left? a. a decline in the stock market b. an increase in taxes c. a decrease in government spending d. all of the above
-
On January 1, 2013, Gandy Corp. sold $600,000 of its own 8 percent, 10-year bonds. Interest is payable annually on December 31. The bonds were sold to yield an effective interest rate of 9percent....
-
Show that every group G with identity e and such that x * x = e for all x G is abelian.
-
A pitot-static tube is inserted into a pipe carrying methyl alcohol at 25C. A differential manometer using mercury as the gage fluid is connected to the tube and shows a deflection of 225 mm....
-
A flow nozzle similar to that shown in Fig. 15.4 is used to measure the flow of water at 120F. The pipe is 6-in Schedule 80 steel. The nozzle diameter is 3.50 in. Determine the pressure difference...
-
Select a long-throated flume from Table 14.5 that will carry a range of flow from 50 m 3 /h to 180 m 3 /h. Compute the head for each of these flows and then compute the flow that would result from...
-
Bramble Corp. constructed a building at a cost of $13900000. Weighted-average accumulated expenditures were $5680000, actual interest was $566000, and avoidable interest was $286000. If the salvage...
-
In 2022, Australias GDP grew very fast while supposing Japans GDP remained constant. Assuming other things are fixed, please use appropriate diagrams or theories to predict the movements of the...
-
Is there anything comparable in US Economic History that we can compare to our current economy? Or are we in a unique economic existence for the first time in our history? Consider these economic...

Study smarter with the SolutionInn App


