A buffer with a pH of 9.85 contains CH 3 NH 2 and CH 3 NH 3
Question:
A buffer with a pH of 9.85 contains CH3NH2 and CH3NH3Cl in water. What can you conclude about the relative concentrations of CH3NH2 and CH3NH3Cl in this buffer? For CH3NH2, pKb = 3.36.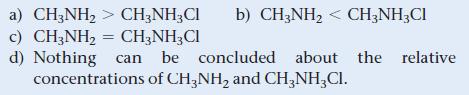
Transcribed Image Text:
a) CH3NH₂ > CH3NH3Cl b) CH3NH₂ < CH3NH3Cl c) CH3NH₂ = CH3NH₂Cl d) Nothing can be concluded about the relative concentrations of CH3NH₂ and CH3NH₂ Cl.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
b C...View the full answer

Answered By

Michael Owens
I am a competent Software Engineer with sufficient experience in web applications development using the following programming languages:-
HTML5, CSS3, PHP, JAVASCRIPT, TYPESCRIPT AND SQL.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A medical researcher wishes to determine the percentage of females who take vitamins. He wishes to be 99% confident that the estimate is within 2% percentage points of the true proportion. A recent...
-
What can you conclude about the relative risk of investing in the United States versus Japan from Figure 7.4?
-
In this problem, we consider the analysis of the combined information from both raters on the shoulder flexion (SF) scores in the posture measurement study. Thus, the questions below concern the data...
-
Use the Principle of Induction to prove the formula for all natural numbers \(n\). \(1+2+3+\cdots+n=\frac{n(n+1)}{2}\)
-
How are allocated joint costs treated when making a sell-or-process-further decision?
-
The state of Missouri has three major power-generating companies (A, B, and C). During the months of peak demand, the Missouri Power Authority authorizes these companies to pool their excess supply...
-
What set of eco-efficiency characteristics is the shareholder value approach concerned with identifying?
-
BatCo makes metal baseball bats. Each bat requires 1 kg. of aluminum at $ 18 per kg. and 0.25 direct labor hours at $ 20 per hour. Overhead is assigned at the rate of $ 40 per labor hour. What...
-
Section 3: Professional Competitive Advantage Describe how the material covered in organizational communication might help you become a more competitive professional. Provide two specific examples...
-
Use the HendersonHasselbalch equation to calculate the pH of a buffer solution that is 0.50 M in NH 3 and 0.20 M in NH 4 Cl. For ammonia, pK b = 4.75.
-
A 1.0-L buffer solution contains 0.100 mol HC 2 H 3 O 2 and 0.100 mol NaC 2 H 3 O 2 . The value of Ka for HC 2 H 3 O 2 is 1.8 * 10 -5 . Because the initial amounts of acid and conjugate base are...
-
Describe the relationship between light wavelength and color vision.
-
Given that f ( x ) = x 2 4 and g ( x ) = 3 x + 5 , find the value of each of the following, if it exists: ( a ) ( f + g ) ( 5 ) ( b ) ( f + g ) ( 1 ) ( c ) ( f g ) ( 0 ) ( d ) ( f g ) ( 2 ) ( e ) ( f...
-
. Assume that over the past 88 years, U.S. Treasury bills had an average return of 3.5% as compared to 6.1% on long-term government bonds. What was the average risk premium on the long-term...
-
The following symbol represents a four-digit up-counter, with synchronous loading function. Complete the schematic for a modulo-12 counter, with this counter and a few gates (AND, OR NOT). The...
-
A particle confined to motion along the x axis moves with constant acceleration from x = 2.0 m to x = 8.0 m during a 2.5-s time interval. The velocity of the particle at x = 8.0 m is 2.8 m/s. What is...
-
Shown in the table is the income of an individual with corresponding unit of good the person can purchase. Fill in the blanks and solve for the income elasticity and to indicate the classification of...
-
What is the difference between an operating and a capital lease?
-
The population of Detroit, Michigan, decreased from 1,027,974 in 1990 to 688,701 in 2013 (Source: U.S. Census Bureau). Find the average rate of change in the population of Detroit, Michigan, over the...
-
Using Fig. 10.87 , design a problem to help other students better understand the superposition theorem. R2 jXL ll V2 -jXc R1 V, (+1
-
Find v o for the circuit in Fig. 10.86 , assuming that i s (t) = 2 sin (2t) + 3 cos (4t) A. i,(t) 10 5 HE Vo rell
-
Determine V o and I o in the circuit of Fig. 10.80 using mesh analysis. J4 u . 2 3V. 2 10/-30 A + >I
-
Let 120) ==== ( cos(0) e sin(0) be a normalized state in C. Is p = |) (4] a density matrix? (20)
-
Using this data on the 2022 annual report, prepare a schedule that calculates goodwill with the reference to Acquisition-Date Fair-Value Allocation Schedule UNITEDHEALTH GROUP CONDENSED CONSOLIDATED...
-
Grocery prices tend to play a role in how people view inflation because of how frequent these purchases are for households. In the past four years grocery prices have jumped 25% which passes overall...

Study smarter with the SolutionInn App


