A chemist in an imaginary universe, where electrons have a different charge than they do in our
Question:
A chemist in an imaginary universe, where electrons have a different charge than they do in our universe, performs the Millikan oil drop experiment to measure the electron’s charge.
The charges of several drops are recorded here. What is the charge of the electron in this imaginary universe?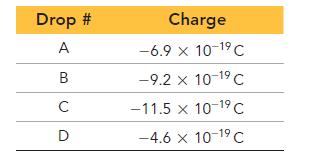
Transcribed Image Text:
Drop # A B C D Charge -6.9 x 10-1⁹ C -9.2 x 10-1⁹ C -11.5 x 10-1⁹ C -4.6 x 10-1⁹ C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The charge of the electron in the imaginary universe is 22 10 z To find the charge ...View the full answer

Answered By

Isaiah Mutinda
As a graduate with Bs in Maths and Computer Science and having worked as a freelance full stack software developer for 3 years running I believe I have what it takes to conformable tutor and mentor a student to a professional developer also.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In the Millikan oil drop experiment, an atomizer (a sprayer with a fine nozzle) is used to introduce many tiny droplets of oil between two oppositely charged parallel metal plates. Some of the...
-
This problem illustrates the ideas behind the Millikan oil drop experiment-the first measurement of the electron charge. Millikan examined a fine spray of spherical oil droplets falling through air;...
-
The Millikan Oil-Drop Experiment The charge of an electron was first measured by the American physicist Robert Millikan during 1909-1913. In his experiment, oil is sprayed in very fine drops (around...
-
Freddie and Jason have just opened the Texas Toothpick, a chain-saw sharpening and repair service located on Elm Street. The Texas Toothpick promises same-week repair service. Freddie and Jason are...
-
How does the total contribution margin differ from the gross margin that is often shown on companies financial statements?
-
Refer tot the annual report for CVS Corporation and the financial statements for Southwest Airline Co. in the Supplement to Chapter 5. Compute the 2008 and 2007 book value per share for both...
-
Claire Hunter plans on opening Claire Hunter Floral Designs. She is considering the various types of business organizations and wishes to organize her business with unlimited life and limited...
-
1. Which principles of globalization did NR and his colleagues follow when they were growing Infosys into a global powerhouse? 2. According to Hofstedes research, India has a high score on the power...
-
FC.61Intermount Wood Products produces finished wood products made from various materials. One of their newer products, reclaimed red oak flooring, is fast becoming a best seller. However, the...
-
As of December 31, 2016, Moss Company had total cash of $195,000, notes payable of $90,500, and common stock of $84,500. During 2017, Moss earned $42,000 of cash revenue, paid $24,000 for cash...
-
Which statements are inconsistent with Rutherfords nuclear theory as it was originally stated? Why? a. Since electrons are smaller than protons and since a hydrogen atom contains only one proton and...
-
Which statements are consistent with Rutherfords nuclear theory as it was originally stated? Why? a. The volume of an atom is mostly empty space. b. The nucleus of an atom is small compared to the...
-
On January 1, Alan King decided to transfer an amount from his checking account into an investment account that later will provide $ 80,000 to send his son to college (four years from now). The...
-
Ahmed was an Arizona University student and spent 20 hours each week working at the university library. He liked the library because it was quiet and he could study some of the time. One interesting...
-
Steven, a junior at Northeast State, just started working part-time at a local fast food restaurant chain. Although not his dream job, it paid for tuition and books, and the restaurant gave him the...
-
Daniel just graduated from Michigan University and landed a job as a copywriter at Young, Olsen, Lindle, and Olson (YOLO) Advertising assigned to one of the subsidiary accounts of Delicious Uber...
-
With the above considered, it is no surprise that Wells Fargo is struggling to keep customers. Despite taking credit for the scandal, having the CEO step down, and implementing marketing campaigns...
-
Monsanto faces challenges that it must address, including lingering concerns over the safety and the environmental impact of its products. The company needs to enforce its code of conduct effectively...
-
Explain why functional currency should be re-measured, rather than translated, when a foreign entitys functional currency is highly inflationary.
-
The water in tank A is at 270 F with quality of 10% and mass 1 lbm. It is connected to a piston/cylinder holding constant pressure of 40 psia initially with 1 lbm water at 700 F. The valve is opened,...
-
Using acetylene as your only source of carbon atoms, identify a synthetic route for the production of 2-bromobutane.
-
Consider the following S N 2 reaction: (a) Draw the mechanism of this reaction. (b) What is the rate equation of this reaction? (c) What would happen to the rate if the solvent is changed from DMSO...
-
Compound A has molecular formula C 7 H 15 Br. Treatment of compound A with sodium ethoxide yields only one elimination product (compound B) and no substitution products. When compound B is treated...
-
Complete this form and upload into the assignment. of Account to Code Patient Acct # Disch Date A / R Days Total Charges Reason Code 2 3 5 9 6 0 0 XX / XX / XX 2 $ 2 3 5 , 6 5 4 . 3 3 2 , 1 2 3 5 9...
-
Thayer Farm trust made a farmer a loan of $1, 200 at 16% for three years compounded annually. Find the future value and the compound interest paid on the loan. Compare the compound interest with...
-
What types of questions would you ask employees and other managers as you conduct a product performance analysis? Provide two examples and explain why answers to these questions would aid in your...

Study smarter with the SolutionInn App


