A mixture of A and B contains a total of 5.3 mols. Both A and B react
Question:
A mixture of A and B contains a total of 5.3 mols. Both A and B react with Z according to the following equations: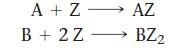
The reaction of the mixture of A and B with Z consumes 7.8 mol Z.
Assuming the reactions go to completion, how many moles of A does the mixture contain?
Transcribed Image Text:
A+Z- B + 2 Z AZ BZ2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
2...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
Ace Ladders has fewer units in beginning finished goods inventory than in ending finished goods inventory. The number of units sold is O greater than the number of units in ending finished goods...
-
The article "Estimating Resource Requirements at Conceptual Design Stage Using Neural Networks" (A. Elazouni, I. Nosair, et al., Journal of Computing in Civil Engineering, 1997:217-223) suggests that...
-
Presented below is information related to LeBron James Manufacturing Corporation. Instructions (a) Compute the rate of depreciation per year to be applied to the plant assets under the composite...
-
Bartlett's Pears has a profit margin of 7.5 percent on sales of $26,000,000. If the firm has debt of $9,500,000 and total assets of $19,000,000, what is the firm's ROA?
-
In flow over a surface velocity and temperature profiles are of the forms u(y) = Ay + By2 Cy3 and T(y) = D + Ey + Fy2- Gy3 where the coefficients A through G are constants. Obtain expressions for...
-
1. Identify 3 students to play the roles of the employees. Ask these 3 individuals to read their roles below. 2. Identify 1 student to play the role of the president of the social enterprise (Taylor...
-
Described below are certain transactions of Edwardson Corporation. The company uses the periodic inventory system. 1. On February 2, the corporation purchased goods from Martin Company for $70,000...
-
Your client, Gabe, needs to calculate the business part of his real estate taxes. How can Gabe calculate the business part of his real estate taxes? Unset starred question Add the real estate taxes...
-
18 The following audit procedures were performed in the audit of inventory to satisfy specific balance-related audit objectives. The audit procedures assume that the auditor has obtained the...
-
A particular kind of emergency breathing apparatusoften placed in mines, caves, or other places where oxygen might become depleted or where the air might become poisonedworks via the following...
-
A mixture of 20.6 g of P and 79.4 g of Cl 2 reacts completely to form PCl 3 and PCl 5 as the only products. Find the mass of PCl 3 that forms.
-
Suppose that you buy a TIPS (inflation-indexed) bond with a 1-year maturity and a coupon of 4% paid annually. If you buy the bond at its face value, and the inflation rate is 8%: a. What will be your...
-
The use of a corporation can provide income tax deferral only if the shareholder(s) can afford to leave the after-tax income in the corporation. Question content area bottom Part 1 A. False because...
-
Because hedge funds are only available to sophisticated investors Question 27 options: they do not need to comply with all of the reporting requirements. they are conservative with investments....
-
Mountaineer Outfitters, a fictitious company, has locations in Asheville, NC; Lexington, KY; and Charlottesville, VA. Mountaineer's management wants to evaluate and compare performance at each of the...
-
shorten and reword the following Selling a property is one of the biggest financial transactions you'll make, at OBrien Real Estate we aim to take the stress out of the equation and deliver a result...
-
Caso 2 Hulera de Sula S. A. es una empresa dedicada a la importacin de hule natural y sinttico para la produccin de llantas y la fabricacin de diversos productos de hule; su planta est ubicada en la...
-
Kearney Company, operating at full capacity, sold 400,000 units at a price of $246.60 per unit during 2012. Its income statement for 2012 is as follows: The division of costs between fixed and...
-
Clark, PA, has been engaged to perform the audit of Kent Ltd.s financial statements for the current year. Clark is about to commence auditing Kents employee pension expense. Her preliminary enquiries...
-
Solid methanol in thermal contact with the surroundings is reversibly melted at the normal melting point at a pressure of 1 atm. Are S, S surroundings , and S total positive, negative, or zero?...
-
A compound with molecular formula C 9 H 10 O exhibits the following spectra ( 1 H NMR, 13 C NMR, and IR). Identify the structure of this compound.. Proton NMR 10 Chemical Shift (ppm) Carbon NMR 128.5...
-
Can incandescent lighting be regarded as an example of cogeneration during the heating season? In a season where air conditioning is required?
-
Estimated total machine-hours used Estimated total fixed manufacturing overhead Estimated variable manufacturing overhead per machine-hour Molding 2,500 $ 12,000 $ 2.20 Fabrication 1,500 $ 16,200 $...
-
Write a MATLAB program that asks the user to enter his results in four quizzes (q1,q2, q3, and q4) out of 100, and calculate the average value. Then print the output given in the Table below to the...
-
Student Name: Anthony Jedruczek (Please PRINT your name) Assume that Q-Caf has the following transactions related to the sale of coffee beans during the month of October, 2023. Oct 1 Oct 5 Oct 15 Oct...

Study smarter with the SolutionInn App


