An aluminum sphere contains 8.55 * 10 22 aluminum atoms. What is the spheres radius in centimeters?
Question:
An aluminum sphere contains 8.55 * 1022 aluminum atoms. What is the sphere’s radius in centimeters?
The density of aluminum is 2.70 g/cm3.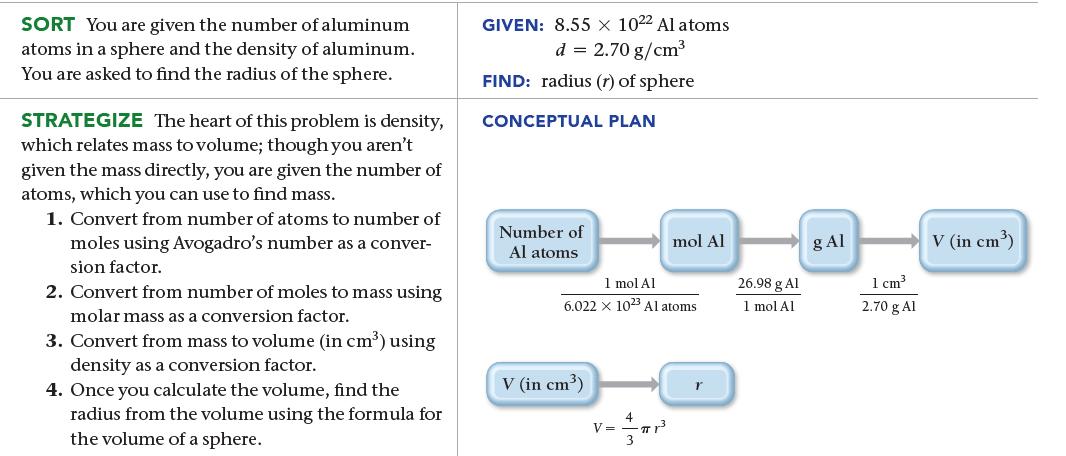


Transcribed Image Text:
SORT You are given the number of aluminum atoms in a sphere and the density of aluminum. You are asked to find the radius of the sphere. STRATEGIZE The heart of this problem is density, which relates mass to volume; though you aren't given the mass directly, you are given the number of atoms, which you can use to find mass. 1. Convert from number of atoms to number of moles using Avogadro's number as a conver- sion factor. 2. Convert from number of moles to mass using molar mass as a conversion factor. 3. Convert from mass to volume (in cm³) using density as a conversion factor. 4. Once you calculate the volume, find the radius from the volume using the formula for the volume of a sphere. GIVEN: 8.55 x 10²2 Al atoms d = 2.70 g/cm³ FIND: radius (r) of sphere CONCEPTUAL PLAN Number of Al atoms 1 mol Al 6.022 x 1023 Al atoms V (in cm³) V = mol Al πTr³ 26.98 g Al 1 mol Al g Al 1 cm³ 2.70 g Al V (in cm³)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
855 x 1022 Al atoms X 4 3 V ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
What is the company' s sustainable growth rate? Suppose the firm targets a 30% sales growth rate next year, and the firm doesn't want to issue new equity, what will be the the firm's new D/A ratio?...
-
The Ebitts Field Corp. manufactures baseball gloves. Charlie Botz, the companys top salesman, has recommended expanding into the baseball bat business. He has put together a project proposal...
-
On July 1,the first day of their fiscal year, the City of Denver sold bonds with a face value of $10,000,000 at 102 percent par. The bonds bear annual interest at 6 percent; interest is payable...
-
Cede & Co. can borrow at 9 percent. Cede currently has no debt, and the cost of equity is 16 percent. The current value of the firm is $540,000. What will the value be if Cede borrows $110,000 and...
-
Dillman Labs, Inc. provides mad cow disease testing for both state and federal governmental agricultural agencies. Because the companys customers are governmental agencies, prices are strictly...
-
Construct an environmental profile for a company of your choice. How might the profile be of use to management?
-
Consider the Emarpy Appliance situation in Problem 6-54. If Richard Feehan wants to minimize the total annual inventory cost, how many refrigerators should be produced in each production run? How...
-
Pina Company's income statement for the year ended December 31, 2025, contained the following condensed information. Service revenue Operating expenses (excluding depreciation) $621,000 Depreciation...
-
Aman and Juanita have $150,000 in their business bank account. You have calculated their warehouse stock and equipment to be valued at a total of $480,000. They want to take out a policy for...
-
Write the symbol for each element and classify it as a metal, nonmetal, or metalloid. a. Gold b. Fluorine c. Sodium d. Tin e. Argon
-
A mixture of CaCO 3 and (NH 4 ) 2 CO 3 is 61.9% CO 3 by mass. Find the mass percent of CaCO 3 in the mixture.
-
Determine the acceleration of block A when the system is released from rest. The coefficient of kinetic friction and the weight of each block are indicated. Neglect the mass of the pulleys and cord....
-
Find and watch videos of three different MVS applications on YouTube or other video-sharing sites. To help you get started, try a couple of the search phrases are listed below. Simply go to YouTube...
-
Assess whether there is an American culture and its characteristics.
-
Small and medium enterprises (SMEs) have played a major role in economic growth across the globe. They are major contributors to national economies in terms of their contribution to employment,...
-
Examine the role of indigenous people in modern-day nations and the challenges that confront them.
-
Are education programs likely to be effective in controlling world population growth? What types of policies may increase or decrease population growth?
-
We talked about concurrent engineering as an alternative to sequential development. What are the advantages of concurrent engineering? Under what circumstances might sequential development be...
-
d) For die casting processes: 1. What are the most common metals processed using die casting and discuss why other metals are not commonly die casted? 2. Which die casting machines usually have a...
-
Acid-catalyzed hydration of 1-methylcyclohexene yields two alcohols. The major product does not undergo oxidation, while the minor product will undergo oxidation. Explane.
-
Calculate S surroundings and S total for part (c) of Problem P5.6. Is the process spontaneous? The state of the surroundings is T = 310.K, P = 0.333 bar.
-
Acid-catalyzed hydration of 1-methylcyclohexene yields two alcohols. The major product does not undergo oxidation, while the minor product will undergo oxidation. Explane.
-
As a long-term investment, Painters' Equipment Company purchased 20% of AMC Supplies Inc's 470,000 shares for $550,000 at the beginning of the fiscal year of both companies. On the purchase date, the...
-
Review both links above about race and U.S. immigration law and policy. Reflecting on how racism has penetrated the treatment of immigrants in the U.S. How might the recent/current public awareness...
-
Explain how occupational health and safety can be linked to 3 specific human resource functions when a company decides to adopt a more strategic approach to OH&S.?

Study smarter with the SolutionInn App


