An oxide of titanium crystallizes with the unit cell shown here (titanium = gray; oxygen = red).
Question:
An oxide of titanium crystallizes with the unit cell shown here (titanium = gray; oxygen = red). What is the formula of the oxide?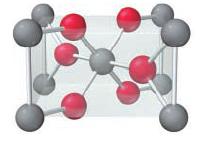
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

Tamil Elakkiya Rajendran
I'm currently involved in the research in the field of Biothermodynamics, Metabolic pathway analysis and computational Biology. I always prefer to share my knowledge whatever I have learnt through my degree whenever time permits.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An oxide of rhenium crystallizes with the unit cell shown here (rhenium = gray; oxygen = red). What is the formula of the oxide?
-
Tungsten crystallizes in the unit cell shown here. (a) What type of unit cell is this? (b) How many tungsten atoms occur per unit cell? (c) If the edge of the unit cell is 316.5 pm, what is the...
-
The unit cell shown here is for calcium carbide. How many calcium atoms and how many carbon atoms are in each unit cell? What is the formula of calcium carbide? (Calcium ions are silver in color and...
-
Starting with acetylene as your only source of carbon atoms, identify how you would prepare each member of the following homologous series of aldehydes: a. Ethanal b. Propanal c. Butanal d. Pentanal
-
St. Charles Drilling Co. contracted with Osterholt to install a well and water system that would produce a specified quantity of water. The water system failed to meet its warranted capacity, and...
-
Assume the following information: Spot rate of Mexican peso ..........= $.100 180-day forward rate of Mexican peso .....= $.098 180-day Mexican interest rate ..........= 6% 180-day U.S. interest rate...
-
Determining the limitations of the constitutional doctrine of due process has been a difficult issue to resolve. Use the Internet to research specific California cases in which a court considered...
-
The following information was summarized from the balance sheets of McDonalds Corporation at December 31, 2008, and Wendys/Arbys Group, Inc., at December 28, 2008. Required 1. Using the information...
-
The funding of DMO's & CVB's in the United Kingdom (UK) differs greatly from the USA. Briefly describe the difference and then say whether, in your opinion, it is better or worse than the USA.
-
The unit cells for cesium chloride and barium chloride are shown here. Show that the ratio of cations to anions in each unit cell corresponds to the ratio of cations to anions in the formula of each...
-
Which solid in each pair has the higher melting point and why? a. Fe(s) or CCl4(s) C. Ti(s) or Ne(s) b. KCI(s) or HCl(s) d. HO(s) or HS(s)
-
Segment PQ has the given coordinates for one endpoint P and for its midpoint M. Find the coordinates of the other endpoint Q. P(2.5, 1.75), M(3, 2)
-
A candy company needs to purchase a new piece of manufacturing equipment. Vendor 1 has equipment that is priced at $150,000, has a capacity of 100 tons per day, and uses $1,000 in power consumption...
-
What is the inventory turnover for 2020? The following information is available for Marigold Company: 2020 2019 Accounts receivable $360000 $333000 Inventory 300000 400000 Net credit sales 3050000...
-
Can you elucidate the role of financial institutions as catalysts for inclusive economic growth and development, examining their contributions to financial inclusion, small and medium-sized...
-
Fleet Street Incorporated a manufacturer of high-fashion clothing for women, is located in South London in the UK. Its product line consists of trousers (28%), skirts (34%), dresses (15%), and other...
-
How can Cost-push inflation be explained ? Explain.
-
fElm Furniture Company, a medium-sized, publicly traded manufacturer of wood-based office and home furniture systems, has agreed that its major goal should be to Become recognized as a value and...
-
Consider model (9.18). What is the effect on the model parameter estimates, their standard errors, and the goodness-of-fit statistics when (a) The times at risk are doubled, but the numbers of deaths...
-
Determine the mean free path for Ar at 298 K at the following pressures: a. 0.500 atm b. 0.00500 atm c. 5.00 10 6 atm For Ar, = 3.6 10 19 m 2 (see Table 33.1) and M = 0.040 kg mol 1 .
-
Determine the mean free path at 500. K and 1 atm for the following: a. Ne b. Kr c. CH 4 Rather than simply calculating the mean free path for each species separately, instead develop an expression...
-
Consider the diagram of a molecular beam apparatus provided in the text. In the design of the apparatus, it is important to ensure that the molecular beam effusing from the oven does not collide with...
-
You decide to take out a 30-year mortgage loan to buy the home of your dreams. The home's purchase price is RM120,000. You manage to secure a RM20,000 down payment and plan to borrow the balance of...
-
Using your computer or podcast player listen to"Reasonable Suspicion" from The Briefing Room podcast. Today's briefing is aboutTerry v. Ohio, the 1968 U.S. Supreme Court case that set the standards...
-
AWM Construction Co. began operations in 2021. Construction activity for 2021 is shown below. AWM uses the cost-recovery method. Billings Collections Estimated Contract Contract Through Through Price...

Study smarter with the SolutionInn App


