Balance each chemical equation. SiO (s) + Ca(HCO3)2(aq) CoS3(s) + NH4NO3(aq) a. CO(g) + CaSiO3(s) + HO(1)
Question:
Balance each chemical equation.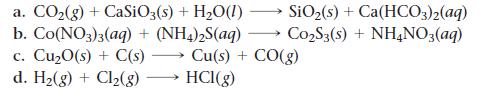
Transcribed Image Text:
SiO₂ (s) + Ca(HCO3)2(aq) Co₂S3(s) + NH4NO3(aq) a. CO₂(g) + CaSiO3(s) + H₂O(1) b. Co(NO3)3(aq) + (NH4)2S(aq) c. Cu₂0(s) + C(s) Cu(s) + CO(g) d. H₂(g) + Cl₂(g) HCI(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a 2 COg CaSiO3s HO1 SiO ...View the full answer

Answered By

AJIN KURIAKOSE
I HAVE ELECTRONICS ENGINEERING DEGREE..AND MY AREA OF INTEREST IS MATHEMATICS,CONTROL SYSTEM,NETWORK,DIGITAL
4.70+
21+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In Exercises 1-3, balance the chemical equation for each reaction.
-
The scatterplot shows the median weekly earning (by quarter) for men and women in the United States for the years from 2005 through 2017. The correlation is 0.974. a. Use the scatterplot to estimate...
-
Total current assets TOTAL ASSETS SHAREHOLDERS' EQUITY AND LIABILITIES: Shareholders' Equity: Preferred stock Common stock: Tk. 10 par 100,000 shares Share Premium 1690 2070 3060 3520 100 100 1000...
-
Consider the following propositions: p: 2 is the smallest prime q: 6 is a perfect square Represent the proposition "2 is the smallest prime but 6 is not a perfect square" using logical connectives....
-
In the table, find the Treasury bond that matures in May 2033. What is the asked price of this bond in dollars? If the bid-ask spread for this bond is two ticks, what is the bid price in dollars?
-
The core of a high-temperature, gas-cooled nuclear reactor has coolant tubes of 20-mm diameter and 780-mm length. Helium enters at 600 K and exits at 1000 K when the flow rate is 8 x 10-3 kg/s per...
-
Mrs. Clarks Foods was an Iowa company engaged in the business of distributing juice beverages. International Suntrade and Miller & Smith Foods were Canadian companies that acted as brokers...
-
(Issuance of Bonds between Interest Dates, Straight-Line, Retirement) Presented below are selected transactions on the books of Simonson Corporation. May 1, 2010 Bonds payable with a par value of...
-
16. A general partnership operates a small resort area in Florida. Jeb is given the opportunity to receive a capital interest of $500 in the partnership in exchange for managing the resort from...
-
John and Barbara were first cousins living in Washington. Under Washington law, they were not permitted to marry. They traveled to Nashville, Tennessee, one weekend and married in a courthouse...
-
Write the balanced chemical equation for the reaction of aqueous potassium hydroxide with aqueous iron(III) chloride to form solid iron(III) hydroxide and aqueous potassium chloride.
-
Write the balanced chemical equation for the reaction of aqueous sodium carbonate with aqueous copper(II) chloride to form solid copper(II) carbonate and aqueous sodium chloride.
-
Do you remember the earlier study by Katz et al. that had students answer SAT-type questions without first reading the passage? (If not, look at Exercises 3.1 and 4.1.) Suppose that we gave out the...
-
Compare the organizational structure of law enforcement agencies with the military including examples from both local law enforcement agencies, as well as state or federal law enforcement agencies...
-
How are cost drivers chosen? What happens to direct costs when a subunit is eliminated?
-
Which database is used by Blockchain? a . . Oracle b . . Big Data c . . MySQL d . . MongoDB
-
What is the type of Consensus in blockchain? a . . Proof of Work ( ( PoW ) ) b . . Proof of Agreement ( ( PoA ) ) c . . Proof of Truth ( ( PoT ) ) d . . Proof of Evidence ( ( PoE ) )
-
Your IT team is tasked to review the requirements to design a security operations center for your company. What is involved in collecting requirements for this project? Why is it often difficult to...
-
Kevin Clavin works at the drive-through window of Big Bad Burgers. Occasionally, when a drive-through customer orders, Kevin fills the order and pockets the customers money. He does not ring up the...
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
Consider the reaction FeO(s) + CO(g) Fe(s) + CO 2 (g) for which K P is found to have the following values: a. Calculate ÎG o R , ÎS o R , and ÎHR???? for this reaction at...
-
If K P is independent of pressure, why does the degree of dissociation in the reaction Cl 2 (g) 2Cl(g) depend on pressure?
-
How does the total number of moles in the reaction system change as T increases? H 2 (g) + Cl 2 (g) 2HCl(g) at equilibrium. Assume ideal gas behavior.
-
A small electric immersion heater is used to heat 87 g of water for a cup of instant coffee. The heater is labeled "120 watts" (it converts electrical energy to thermal energy at this rate)....
-
Oxygen gas having a volume of 1250 cm 3 at 43.1 C and 1.04 x 10 5 Pa expands until its volume is 1900 cm 3 and its press is 1.08 x 10 5 Pa. Find: (a) the number of moles of oxygen present and (b) the...
-
An ATV has an average acceleration of 1.9 m/s 2 . If the vehicle accelerates for 5.8 s and has an initial velocity of 15 m/s [E], what is the final velocity of the ATV?

Study smarter with the SolutionInn App


