Common aspirin is acetylsalicylic acid, which has the structure shown here and a pK a of 3.5.
Question:
Common aspirin is acetylsalicylic acid, which has the structure shown here and a pKa of 3.5.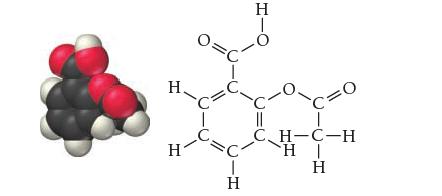
Calculate the pH of a solution in which one normal adult dose of aspirin (6.5 * 102 mg) is dissolved in 8.0 ounces of water.
Transcribed Image Text:
Η. H Η H c=0 CH-C-H H | Η · Η
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The active ingredient in aspirin is acetylsalicylic acid (HC9H7O4), a monoprotic acid with Ka = 3.3 ( 10-4 at 25 (C. What is the pH of a solution obtained by dissolving two extra-strength aspirin...
-
Acetylsalicylic acid (aspirin) has the structure shown by the following molecular model: Describe the geometry and bonding about the carbon atom shown at the top of the model, in which the carbon...
-
The active ingredient in aspirin is acetylsalicylic acid (HC 9 H 7 O 4 ), which has a K a of 3.00 x 10 -4 . A 325-mg tablet of aspirin is dissolved in 25.00 mL of water, and the solution is titrated...
-
Write a program RecoverSignal that will read the binary file written by StoreSignal, as described in the previous exercise. Display the integer values that the data represents on the screen.
-
Refer to the data in Exercise 9-4. Required: 1. Compute overhead variances using a two-variance analysis. 2. Compute overhead variances using a three-variance analysis. 3. Illustrate how the two- and...
-
An annual report for Ford Motor Company included the following information: Note 6. Net Property, Depreciation and Amortization-Automotive Assets placed in service before January 1, 1993, are...
-
How to obtain an extension of time and continuances?
-
Comparing Companies within an Industry Refer to the financial statements of American Eagle Outfitters (Appendix B) and Urban Outfitters (Appendix C) and the Industry Ratio Report (Appendix D) at the...
-
Research an association that you'd like to join. Report back to us the website (URL) and be sure to at leastanswer the questions: 1. Why would you want to belong to this association, 2. What do they...
-
White wines tend to be more acidic than red wines. Find the [H 3 O + ] in a Sauvignon Blanc with a pH of 3.23 and a Cabernet Sauvignon with a pH of 3.64. How many times more acidic is the Sauvignon...
-
Acid rain over the Great Lakes has a pH of about 4.5. Calculate the [H 3 O + ] of this rain and compare that value to the [H 3 O + ] of rain over the West Coast that has a pH of 5.4. How many times...
-
If you could choose between what we called the Anglo-American political economy and the continental European model of political economy, which would you prefer? Why?
-
Using a suitable method, forecast the number of newborns in Singapore for 2020 3Q through 2021 4Q. Please provide justification for the method you choose. Please limit the answer to within two pages.
-
Beginning inventory Hemming Company reported the following current-year purchases and sales for its only product. Date January 1 Activities Units Acquired at Cost 255 units @ $12.20 = $ 3,111 Units...
-
Current Attempt in Progress Kingbird Enterprises owns the following assets at December 31, 2023: $48,200 Chequing account balance $31,000 14,300 Postdated cheque from Blossom Company 450 taxes)...
-
A distant cousin of yours had inherited some money so he was able to realise his life long ambition to set up in business and sell that well known financial publication, the National Review. He put R...
-
Assume a 10% discount rate and compute the present value of Rs. 1100, Rs.900, Rs.1500 andRs.700 received at the end of 1-4 years In the above sum if the amounts are received at the beginning of the...
-
Compute the Z score for Central Manufacturing Corporation (CMC) given the following information for year-end 2012: Based on its Z score, is CMC likely to go bankrupt in the near future? CMC Financial...
-
Activator rod AB exerts on crank BCD a force P directed along line AB. Knowing that P must have a 100-N component perpendicular to arm BC of the crank, determine (a) The magnitude of the force P, (b)...
-
Oil exploration. When searching for gold, measurements of g can be used to find regions within the Earth where the density is larger than that of normal soil. Such measurements can also be used to...
-
The International Space Station orbits at an average height of 350 km above sea level. (a) Determine the acceleration due to gravity at that height and find the orbital velocity and the period of the...
-
An astronaut stands on the surface of Vesta, which, with an average radius of 270 m, makes it the third largest object in the asteroid belt. The astronaut picks up a rock and drops it from a height...
-
a. Futurist Laptop Repairs and Services offer the following services. Write a function named calcServ Price () to determine total price a customer should pay depending on type of service chosen. The...
-
When economists evaluate the fairness of financing health care, they assess potential payment methods such as income tax, deficit financing, sales tax, payroll tax, and user tax. If you could choose...
-
Given below are the statements of profit or loss of two companies for the year ended 31 December x6. Guess RM 2,000,000 Sure RM 1,600,000 Turnover (600,000) (400,000) Cost of sales 1,400,000...

Study smarter with the SolutionInn App


