Complete and balance each equation. If no reaction occurs, write NO REACTION. a. Lil(aq) + BaS(aq) b.
Question:
Complete and balance each equation. If no reaction occurs, write “NO REACTION.”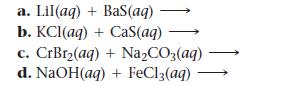
Transcribed Image Text:
a. Lil(aq) + BaS(aq) b. KCl(aq) + CaS(aq) c. CrBr₂(aq) +Na₂CO3(aq) d. NaOH(aq) + FeCl3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a NO REACTION b NO ...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Complete and balance each of the following molecular equations, including phase labels, if a reaction occurs. Then write the net ionic equation. If no reaction occurs, write NR after the arrow. a....
-
Complete and balance each of the following molecular equations, including phase labels, if a reaction occurs. Then write the net ionic equation. If no reaction occurs, write NR after the arrow. a....
-
Complete and balance each of the following equations. If no reaction occurs, write NOREACTION. Express your answer as a chemical equation. Identify all of the phases in your answer. 1) KCl (aq) + CaS...
-
All else equal, a large sell off in the equity markets (decline in the S&P 500 and the Dow Jones Industrial Average) would be the most likely result from the reporting of a much higher than expected...
-
Recall that the salami technique means using a computer to skim a small amount of money from hundreds or thousands of accounts, and then diverting the proceeds for personal gain. Suppose that a...
-
Gallatin Carpet Cleaning is a small, family-owned business operating out of Bozeman, Montana. For its services, the company has always charged a flat fee per hundred square feet of carpet cleaned....
-
Gilead is a large drug producer, with a majority of its prescription drug product sales occurring in the United States. Gilead produces anti-HIV drug therapies, including the drugs Atripla, Truvada,...
-
The city of Merlot operates a central garage through an Internal Service Fund to provide garage space and repairs for all city-owned and -operated vehicles. The Central Garage Fund was established by...
-
A violet ray of light leaves a medium with refractive index of 6.46 at an angle of 0.07 radians to the normal and enters a second medium with refractive index of 22.04. At what angle to the normal...
-
Complete and balance each equation. If no reaction occurs, write NO REACTION. a. NaNO3(aq) b. NaCl (aq) + c. (NH4)2SO4(aq) + SrCl(aq) d. NH4Cl(aq) + AgNO3(aq) + KC1(aq) Hg2(CH3O2)2(aq)
-
Determine whether each compound is soluble or insoluble. If the compound is soluble, list the ions present in solution. a. Agl c. COCO3 b. Cu3(PO4)2 d. K3PO4
-
Calculate the value of a put option with exercise price $100 using the data in Problem 36. Show that put-call parity is satisfied by your solution.
-
A toy store is considering stocking a new toy that has just been released in the market. To test the marketability of the toy, the toy store will place an initial single order for the toys. Each toy...
-
Whether the optimal level of product availability is high or low depends on where a particular company believes they can minimize cost. maximize revenue. maximize profits. maximize product...
-
When is pricing the use of transportation infrastructure, like roads, considered necessary? How is pricing structured?
-
The cost of holding inventory incurred by the shippers supply chain network is transportation cost. inventory cost. facility cost. processing cost.
-
Consider a firm like Zara that has developed production capabilities with very short replenishment lead times. Do you think this capability is more valuable for its online operations or its store...
-
During the current year, Sevcik Company had pretax income from continuing operations of $19,200, which included interest expense of $4,000. At the end of the year, Sevcik Company had total assets of...
-
Refrigerant R-12 at 30C, 0.75 MPa enters a steady flow device and exits at 30C, 100 kPa. Assume the process is isothermal and reversible. Find the change in availability of the refrigerant.
-
Air at 16C and standard atmospheric pressure has a specific weight of 12.02 N/m 3 . Calculate its density.
-
Using the data from Table 2.5, report the minimum, nominal, and maximum values for viscosity for ISO grades VG 10, VG 65, VG 220, and VG 1000. Kinematic Viscosity at 40C (cSt) or (mm2/s) Grade ISO VG...
-
If you were asked to check the viscosity of an oil that is described as SAE 40, at what temperatures would you make the measurements?
-
Describe the Query Processing in a Directed Database Management Systems with the following steps: Query decomposition Data localization Global optimization . Local optimization
-
The purpose of this assignment is to determine if the database performance can be improved by monitoring response times for frequently used queries and determining if the queries were written...
-
Study the index definitions in sh_idx.sql. These indexes have already been created in SH2. Whatever indexes you decide to create for this task should be the result of your own research and thinking...
Beginners Blueprint For Real Estate Investing Success 1st Edition - ISBN: 979-8866680641 - Free Book

Study smarter with the SolutionInn App


