Complete and balance each gas-evolution equation. a. HNO3(aq) + NaSO3(aq) b. HCl(aq) + KHCO3(aq) c. HC2HO(aq) +
Question:
Complete and balance each gas-evolution equation.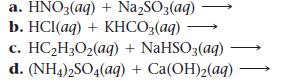
Transcribed Image Text:
a. HNO3(aq) + Na₂SO3(aq) b. HCl(aq) + KHCO3(aq) c. HC₂2H₂O₂(aq) + NaHSO3(aq) d. (NH4)2SO4(aq) + Ca(OH)₂(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The complete and balanced gasevolution equations are as follows a HNO3aq Na2SO3aq H2SO4aq NaNO3aq NO...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Complete and balance each combustion equation. a. C4H9OH + O2 ( ? b. CH3NO2 + O2 ( ?
-
Complete and balance each combustion equation. a. B2H6 + O2 ( ? (The oxide of boron formed is B2O3.) b. Al2S3 + O2 ( ? (The oxide of sulfur formed is SO2.) c. Al2S3 + O2 ( ? (The oxide of sulfur...
-
Complete and balance each of the following molecular equations, including phase labels, if a reaction occurs. Then write the net ionic equation. If no reaction occurs, write NR after the arrow. a....
-
why is teamwork so important especially in healthcare? explain
-
Why is it important for managers to evaluate internal controls?
-
What is everyday low pricing and how does it impact the bullwhip effect?
-
Haupt Consulting, Inc., began operations and completed the following transactions during the first half of December: Requirements 1. Analyze the effects of Haupt Consultings transactions on the...
-
The following information is available for Laurel Company, a wholesale company: Expected sales volume: October . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
On April 1, Hao Lee created a new travel agency, Lee Travel The following transactions occurred during the company's first month April 2 Lee Invested $40,000 cash and computer equipment worth $12,600...
-
Assign oxidation states to each atom in each element, ion, or compound. a. Ag d. HS b. Ag e. CO3- c. CaF f. CrO4- 2-
-
Complete and balance each gas-evolution equation. a. HBr(aq) + NIS(s) b. NH4l (aq) + NaOH(aq) c. HBr(aq) + NaS(aq) d. HCIO4(aq) + LiCO3(aq)
-
In Morgan Company, data concerning two products are: Contribution margin per unitProduct A $11, Product B $12; machine hours required for one unitProduct A 2, Product B 2.5. Compute the Contribution...
-
1. Consider a particle of mass m and mechanical energy & moving in the potential U (x) U cosh(x/a)' where u and a are positive constants. (b) Now consider the motion of a particle incident on the...
-
Use the continuous compound interest formula to find the indicated value. A=$81,000; P= $59,871; r = 9.3%; t = ?
-
Currently, the term structure is as follows: One-year bonds yield 11.00%, two-year zero-coupon bonds yield 12.00%, three-year and longer maturity zero-coupon bonds all yield 13.00%. You are choosing...
-
Consider the parametric curve: x=8+11 cost, y=10+11 sint, The cartesian equation of the curve has the form (x - h) + (y - k) = R with h = k = R = and The initial point has coordinates: x = ,y= The...
-
Daily Oxygen. The density of air under standard temperature and pressure is about 1.29 kg/m. Typically, a person breathes about 0.50 L per breath. Approximately how many kilograms of oxygen (not...
-
Although he was initially satisfied by the apparent profitability of the new project, the Avon company's chief executive officer (CEO) was troubled by some other aspects of the project that he...
-
Dr. Chan obtained a $15,000 demand loan at prime plus 1.5% on September 13 from the Bank of Montreal to purchase a new dental X-ray machine. Fixed payments of $700 will be deducted from the dentists...
-
An oil is tested using a Saybolt viscometer and its viscosity is 526 SUS at 40C. Determine the kinematic viscosity of the oil in mm 2 /s at that temperature.
-
Convert all of the kinematic viscosity data in Table 2.5 for ISO viscosity grades from mm 2 /s (cSt) to SUS. Kinematic Viscosity at 40C (cSt) or (mm2/s) Grade IS VG Nominal Minimum Maximum 2.2 1.98...
-
Convert 1600 square millimeters to square meters.
-
After walking through the fifth gate, your computer informs you that more than half of the gates have been crossed. "Only 4 left", you conclude. "Computer, what is this gate's riddle?" Your computer...
-
Find the derivative of the following function without using the product rule, f ( x ) = 7 x ( x 3 1 1 x 2 + 4 x + 4 ) First, rewrite the function in an equivalent way so that the product rule need...
-
Find the area of the region that is bounded by the given curve and lies in the specified sector. 3 3 r = e, < 0 < 4 2

Study smarter with the SolutionInn App


