Complete and balance each hydrocarbon combustion reaction. a. CHCHCH3 + 0- b. CHCHCH=CH + 0 c. CH=CH
Question:
Complete and balance each hydrocarbon combustion reaction.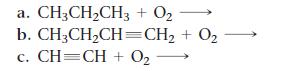
Transcribed Image Text:
a. CH₂CH₂CH3 + 0₂- b. CH₂CH₂CH=CH₂ + 0₂ c. CH=CH + O2 →
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a CHCHCH3 5O ...View the full answer

Answered By

Bree Normandin
Success in writing necessitates a commitment to grammatical excellence, a profound knack to pursue information, and a staunch adherence to deadlines, and the requirements of the individual publication. My background comprises writing research projects, research meta-analyses, literature reviews, white paper reports, multimedia projects, reports for peer-reviewed journals, among others. I work efficiently, with ease and deliver high-quality outputs within the stipulated deadline. I am proficient in APA, MLA, and Harvard referencing styles. I have good taste in writing and reading. I understand that this is a long standing and coupled with excellent research skills, analysis, well-articulated expressions, teamwork, availability all summed up by patience and passion. I put primacy on client satisfaction to gain loyalty, and trust for future projects. As a detail-oriented researcher with extensive experience surpassing eight years crafting high-quality custom written essays and numerous academic publications, I am confident that I could considerably exceed your expectations for the role of a freelance academic writer.
5.00+
7+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Complete and balance each hydrocarbon combustion reaction. a. CH,CH,CH,CH, + O, b. CH=CHCH3 + 02 c. CH=CCHCH3 + O
-
A combustion reaction involves the reaction of a substance with oxygen gas. The complete combustion of any hydrocarbon (binary compound of carbon and hydrogen) produces carbon dioxide and water as...
-
Complete combustion of a hydrocarbon causes the carbon atoms in the hydrocarbon molecule to be oxidised to their highest oxidation state, +4. See the experiment which shows the complete combustion of...
-
In 2020-21, a taxpayer makes a number of disposals, as listed below. Which of these disposals would be exempt from CGT? (a) An antique table sold for 5,000. (b) A watercolour painting sold at...
-
Cameron Company has current assets equal to $250,000. Of these, $135,000 is cash, $55,000 is accounts receivable, and the remainder is inventories. Current liabilities total $200,000. Required: Round...
-
A silver dollar is flipped twice. Calculate the probability of each of the following occurring: (a) A head on the first flip (b) A tail on the second flip given that the first toss was a head (c) Two...
-
True or False: If \(E R R>M A R R\), then MIRR \(>\) MARR.
-
Multiple-choice questions: a. Which of the following statements is incorrect? 1. Ratios are fractions expressed in percent or times per year. 2. A ratio can be computed from any pair of numbers. 3. A...
-
One way in which economic growth is measured apart from GDP is in Purchasing Power Parity (PPP) terms. The Big Mac Index is the PPP concept applied to the cost of a Big Mac in different countries....
-
Draw a structure for each alkane. a. 2,2-dimethylpentane b. 3-isopropylheptane c. 4-ethyl-2,2-dimethylhexane d. 4,4-diethyloctane
-
Draw a structure for each alkane. a. 3-ethylhexane b. 3-ethyl-3-methylpentane c. 2,3-dimethylbutane d. 4,7-diethyl-2,2-dimethylnonane
-
Use the data in MURDER.RAW for this exercise. The variable mrdrte is the murder rate, that is, the number of murders per 100,000 people. The variable exec is the total number of prisoners executed...
-
1. +23 2. 3. Solve for x: 3x + 7 = 22. Find the area of a triangle with base 8 units and height 10 units. Find the derivative of f(x) = 3x+4x-2.
-
Why is income considered a relevant factor in creating a retirement plan?
-
CONTINUOUS DISCLOSURE OBLIGATIONS Can these detailed results be reported? If yes, then explain how. If no, then explain why not. Refer to the ASX Listing Rules, ASX Scoping Studies guidance, ASX FAQs...
-
Provide the data below Answer the questions. Current Account 1970 1980 1985 1990 2000 2009 Exports of goods and services and income 68,387 344,440 387,612 706,975 1,421,515 2,159,000 receipts Exports...
-
The need for change management in the project management processes. How can the application of change management principles impact the project as a whole?
-
Develop a histogram of the time it took for you or your friends to receive six recent orders at a fast-food restaurant.
-
What are the two methods used to translate financial statements and how does the functional currency play a role in determining which method is used?
-
High microwave intensities can cause biological damage through heating of tissue; a particular concern is cataract formation. The U.S. Food and Drug Administration limits microwave radiation near the...
-
Use the fact that sunlight intensity at Earths orbit is 1364 W/m 2 to calculate the Suns total power output.
-
A quasar 10 billion light-years from Earth appears the same brightness as a star 50,000 light-years away. How do the power outputs of quasar and star compare?
-
Please assist in checking an S Corporation 1120S Tax Return. For the form 1120S, I am struggling to reconcile Line 18 on the schedule k with Line 8 on the Schedule M-1. I have provided all relevant...
-
The population of coyotes in a national forest has consistently increased by 20% each year. This year, the population of coyotes is 5,050. If the population increases at the same rate, what number of...
-
Mark and David are two friends who are both planning to buy a new car. They are choosing between two models of Ford's flagship vehicle: the 2017 Ford Fusion SE and the 2017 Ford Fusion Hybrid. The...

Study smarter with the SolutionInn App


