Consider the curve shown here for the titration of a weak base with a strong acid and
Question:
Consider the curve shown here for the titration of a weak base with a strong acid and answer each question.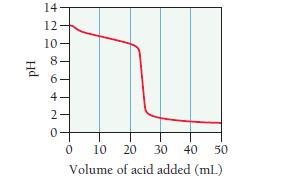
a. What is the pH and what is the volume of added acid at the equivalence point?
b. At what volume of added acid is the pH calculated by working an equilibrium problem based on the initial concentration and Kb of the weak base?
c. At what volume of added acid does pH = 14 - pKb?
d. At what volume of added acid is the pH calculated by working an equilibrium problem based on the concentration and Ka of the conjugate acid?
e. Beyond what volume of added acid is the pH calculated by focusing on the amount of excess strong acid added?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





