Consider the molecular view of an electrochemical cell involving the overall reaction: Draw a similar sketch of
Question:
Consider the molecular view of an electrochemical cell involving the overall reaction:![]()
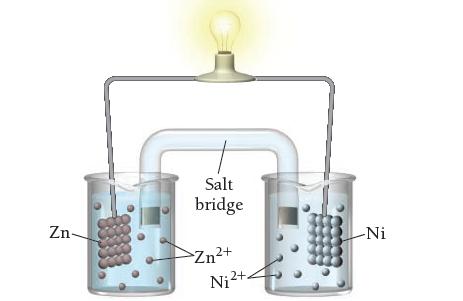
Draw a similar sketch of the cell after it has generated a substantial amount of electrical current.
Transcribed Image Text:
Zn(s) + Ni²+(aq) Zn²+(aq) + Ni(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Zns Ni2aq Zn2aq Nis This is a redox reactionwhere zinc metal is oxidized to zinc ionsand nickel ions ...View the full answer

Answered By

Joseph Ogoma
I have been working as a tutor for the last five years. I always help students to learn and understand concepts that appears challenging to them. I am always available 24/7 and I am a flexible person with the ability to handle a wide range of subjects.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For reaction, A + B 10L volumetric flask at 25C. If K =0.238 are available? C + D: Given that initial amounts of 0.68 mol of both A and B in a at equilibrium, how many moles of each reactant and...
-
The diagram shows an electrochemical cell involving two metal/metal-ion systems. The standard electrode potentials for the half-cells are: Ag + + e Ag E = +0.80 V Cu2 + + 2e Cu E = +0.34 V a....
-
a. Draw a diagram of an electrochemical cell consisting of a Mn 2+ /Mn half-cell and a Pb 2+ /Pb half-cell. b. Use the data in Appendix 2 to calculate the cell voltage. c. Which half-cell is the...
-
Economist Abba Lerner proposed a tariff on oil imports equal to 100 percent of the import price. This tariff is designed to reduce dependence on foreign sources as well as to discourage OPEC from...
-
Sorensen Manufacturing produces several types of bolts used in aircrafts. The bolts are produced in batches according to customer orders. Although there are a variety of bolts, they can be grouped...
-
Debate the relative merits of fixed and floating exchange rate regimes. From the perspective of an international business, what are the most important criteria in a choice between the Critical...
-
In 2013, Thomas DePrince, a passenger aboard a cruise ship, visited the ships jewelry boutique, operated by Starboard Cruise Services, Inc. DePrince told the employees of the boutique that he was...
-
Yellow Press, Inc. buys paper in 1,500-pound rolls for printing. Annual demand is 2,500 rolls. The cost per roll is $800 and the annual holding cost is 15 percent of the cost. Each order costs $50....
-
5. [10] Alice and Bob play a game on a graph G, alternately choosing distinct vertices. Alice starts by choosing any vertex. Each subsequent choice must be adjacent to the preceding choice (of the...
-
Determine whether HI can dissolve each metal sample. If it can, write a balanced chemical reaction showing how the metal dissolves in HI and determine the minimum volume of 3.5 M HI required to...
-
Consider the molecular views of an Al strip and Cu 2+ solution. Draw a similar sketch showing what happens to the atoms and ions after the Al strip is submerged in the solution for a few minutes. Al...
-
Morgan Insurance Ltd. issued a fixed-rate perpetual preferred stock three years ago and placed it privately with institutional investors. The stock was issued at $25.00 per share with a $1.75...
-
compare and contrast HR functions in Brazil with the USA. The HR functions that I am going to compare and contrast are training, human resources development, and performance evaluation. How are they...
-
Suppose you buy one contract for March 2017 delivery at the closing price. If the contract closes in March at a price of $3.99 per bushel, what will be your profit or loss? (Each contract calls for...
-
Let B 4.85 m at 60.0. C and A have equal magnitudes. The direction angle of C is larger than that of A by 25.0. Let A B 27.9 m and B C 33.9 m. Find the magnitude (in m) and direction (in degrees) of...
-
How does criminal justice theories addresses internal organizational challenges in the criminal justice system?
-
Contrast the cultures of the two organizations Hewlett-Packard and Xerox using the following dimensions, be sure to include these dimensions in your answer: Strength of the culture Fit of the culture...
-
Charles Lackey operates a bakery in Idaho Falls, Idaho. Because of its excellent product and excellent location, demand has increased by 25% in the last year. On far too many occasions, customers...
-
Do animals have rights? If so, what are they? What duties do human beings have toward animals? Does KFC protect animal welfare at an acceptable level?
-
A buildup of fission products poisons a reactor, dropping the multiplication factor to 0.992. How long will it take the reactor power to decrease by half, given a generation time of 0.10 s?
-
The total thermal power generated in a nuclear power reactor is 1.5 GW. How much U-235 does it consume in a year?
-
New Hampshires Seabrook nuclear power plant produces electrical energy at the rate of 1.2 GW and consumes 1311 kg of U-235 each year. Assuming the plant operates continuously, find (a) its thermal...
-
Who issues warrants? Multiple choice question. Investment bankers Stock exchanges The firm Stock brokers
-
Substitute the given values into the formula, and solve for the unknown variable. h A==h(B+b); A=20, B=2, b=3
-
Compute the distance between the points. (-10, -22) and (-18,-28) The distance between the given points is (Type an exact answer, using radicals as needed.)

Study smarter with the SolutionInn App


