Consider the reaction: Consider also this representation of an initial mixture of N 2 H 4 and
Question:
Consider the reaction:![]()
Consider also this representation of an initial mixture of N2H4 and N2O4: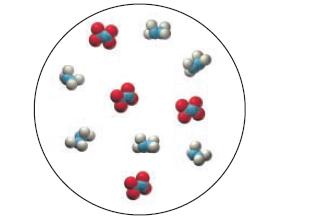
Which diagram best represents the reaction mixture after the reactants have reacted as completely as possible?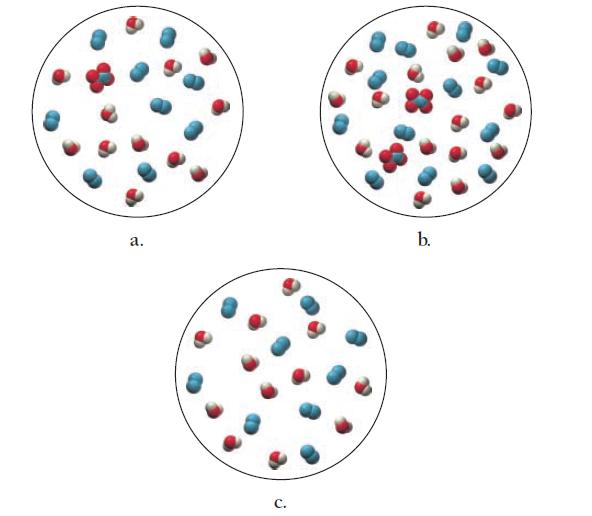
Transcribed Image Text:
2 N₂H4(8) + N₂O4(8) 3 N₂(g) + 4H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the reaction: A reaction mixture initially contains 5 moles of NO and 10 moles of H 2 . Without doing any calculations, determine which set of amounts best represents the mixture after the...
-
Nitrogen and hydrogen gas react to form ammonia according to the reaction: A flask contains a mixture of reactants represented by the image shown at the left. Which of the following images best...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
Solve Prob. 410 using singularity functions. Use statics to determine the reactions. Prob. 410, The figure shows a cantilever consisting of steel angles size 100 Ã 100 Ã 12 mm mounted...
-
Riggins Trucking, Inc., has a current stock price of $41. For the past year, the company had net income of $5,150,000, total equity of $21,580,000, sales of $39,000,000, and 4.1 million shares of...
-
Research each of the following companies and provide three examples of business processes that are likely to be audit-sensitive processes. For each company, provide one process that is critical to...
-
A gas obeys the ideal gas law at (a) Low pressure and high temperature (b) Low pressure and low temperature (c) Low temperature and high pressure (d) High temperature and high pressure.
-
A farmer in Georgia has a 100-acre farm on which to plant watermelons and cantaloupes. Every acre planted with watermelons requires 50 gallons of water per day and must be prepared for planting with...
-
5. Let E(x) = xn which converges for all x. A power series Cnx" can be found for n=0 n=0 E(x) 1+x n!' using power series multiplication. Find Co, C1, C2, C3, C4 of this power series. You do not need...
-
Go to the website www.cedarfair.com and click on Investors, then Investor Information, Financial Reports, and Annual Reports. Then click on 2017 Annual Report on 10-K to access the 2017 Form 10-K...
-
Octane (C 8 H 18 ), a component of gasoline, reacts with oxygen to form carbon dioxide and water. Write the balanced chemical reaction for this process by passing a single piece of paper around your...
-
Hydrolysis of the compound B 5 H 9 forms boric acid, H 3 BO 3 . Fusion of boric acid with sodium oxide forms a borate salt, Na 2 B 4 O 7 . Without writing complete equations, find the mass (in grams)...
-
Show that the given function is a solution of the initial-value problem. dy y = 5e2* + x; - 2y = 1 - 2x, y(0) = 5 dx
-
Assume that you have been invited to ONE of the following social/cultural events for the very first time: 1.a Jewish Passover Seder 2.a Day of the Dead family picnic in a Mexican graveyard 3.a formal...
-
Write to Oriental Bank of Commerce, Civil Lines, Bhopal, to make enquiries about the credit standing of Messrs. Rajpal Brothers, Raja Bazar, Bhopal, for a sum of Rs.5,00,000. The party's bankers are...
-
Discuss how decreasing prices as a way to eliminate competition could result in more competition, and give examples of how competitors could use the price decrease to reduce market share.
-
Based on the feedback you have received on your outline, construct a proper proposal on the course you would like Alpha to offer. Students must: Construct a sequence of logical reasons to support...
-
Can I get an example or a better understanding of how to do one at my organization? I worked for McDonald's as a manager and wanted to know if I could get something explaining how I would go about...
-
What are some of the cultural values of such groups as Hispanics, African-Americans, and Asian Americans?
-
F.(3e* -2x 3 sin(2x)) is equal to 2 3 Cos 8. IT 3, t (4+@ 2 3, 1+o 1 4 Cos 4 4 1 3. 1 +4cos V7 (1+o 4 1 4 Cos 4 1+0 4-
-
Is the following statement true of false? If it is false, rephrase it so that it is true. The entropy of a system cannot increase in an adiabatic process.
-
Which of the following processes is spontaneous? a. The reversible isothermal expansion of an ideal gas. b. The vaporization of superheated water at 102C and 1 bar. c. The constant pressure melting...
-
One joule of work is done on a system, raising its temperature by one degree centigrade. Can this increase in temperature be harnessed to do one joule of work? Explain.
-
On a chilly December morning in Portland, Maine, Jamie decides to visit a local caf owned by Charlie. Overnight, a light snowfall had covered the city, leaving sidewalks slippery. Charlie, aware of...
-
You are a parole officer working on one of your cases. The parolee is waiting for you in the reception area. You read her file and see that the woman is a single mother with three hyperactive,...
-
A point charge q = 4.35 nC is located on the x-axis at x = 1.80 m, and a second point charge q = -6.25 nC is on the y-axis at y = 1.15 m. Part A What is the total electric flux due to these two point...

Study smarter with the SolutionInn App


