Consider this overall reaction, which is experimentally observed to be second order in X and first order
Question:
Consider this overall reaction, which is experimentally observed to be second order in X and first order in Y:![]()
a. Does the reaction occur in a single step in which X and Y collide?
b. Is this two-step mechanism valid?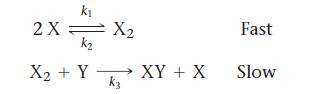
Transcribed Image Text:
X + Y→→→ XY
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a No the reaction does not occur in a single step in ...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider this overall reaction, which is experimentally observed to be second order in AB and zero order in C: Is the following mechanism valid for this reaction? AB + C A + BC
-
Two isomers (A and B) of a given compound dimerize as follows: Both processes are known to be second order in the reactant, and k1 is known to be 0.250 Lmol-1s-1 at 258C. In a particular experiment...
-
A reaction was found to be second order in carbon monoxide concentration. The rate of the reaction _____ if the [CO] is doubled, with everything else kept the same. Please explain. A) remains...
-
Bob Bristol just called to congratulate you on your excellent work on the various assignments at CII.He now wants you to do some capacity analysis for Meghan Willoughby, the Chief Purser. Meghan is...
-
Young, Coopers, and Touche (YCT) is a tax services firm. The firm is located in San Diego and employs 10 professionals and eight staff. The firm does tax work for small businesses and well-to-do...
-
Efendi Company hired an accounting intern, Pat Morgan, to prepare its income statement, statement of retained earnings, and balance sheet. Pat indicated a reluctance to undertake this task due to a...
-
Are there conditions under which a firm might be better off if it were to choose a machine with a rapid payback rather than one with a larger XPV? AppendixLO1
-
On September 15, Krug Company purchased merchandise inventory from Makarov with an invoice price of $ 35,000 and credit terms of 2/10, n/30. Schneider Company paid Makarov on September 28. Prepare...
-
The following trial balance was extracted from the books of Hermas Trading as at 31 December 2020. Particulars Debit (RM) Credit (RM) 298,000 250,000 16.500 2.500 11,000 4,550 9,000 2,300 36,000...
-
Consider this three-step mechanism for a reaction: a. What is the overall reaction? b. Identify the intermediates in the mechanism. c. What is the predicted rate law? k Cl(8) k Cl(g) + CHCl3(8)...
-
From a process, 100 kg of steam is available at 2 MPa and 800C. a. Determine the maximum amount of shaft work that can be obtained from this steam in a non-flow process if the ambient conditions are...
-
Analyze the following accrual- type events, and add them to the work sheet you prepared for P15- 1. 1. Adjust for the expiration of 1 months rent. 2. Adjust for the using up of the furniture during...
-
The goal is to understand the principles of project valuation and capital budgeting in a practical setting, using a combination of fictitious data and real-world examples. Begin by proposing a...
-
45. The use of realistic predetermined unit costs to facilitate product costing, cost control, cost flow, and inventory valuation is a description of the A. flexible budget concept. B. budgetary...
-
2vx Voy Ax g 2vo cos 0 sin 0 g vo sin(20) g
-
PORTAGE COLLEGE Diversity Awareness Course Score | Home | Help | Exit Module 2 Post-Test Module 1 Module 2 Module 3 Module 2 Post-Test Betsy really likes working at Thompson Trucking. She likes how...
-
Suppose f(x) = 5x cos x. Find the equation of the tangent line to f(x) at the point (, -5). y = x+
-
Given the data sets shown in Figures 5.6, explain how the decision tree, naive Bayes, and k-nearest neighbor classifiers would perform on these data sets. Figures 5.6 Attributos Attributos shing...
-
What types of questions can be answered by analyzing financial statements?
-
Many power stations get rid of their waste heat by using it to boil water and allowing the resulting steam to escape into the atmosphere via a cooling tower. How much water would a power station need...
-
How much heat is given off when 1 kg of steam at 100C condenses and cools to water at 20C?
-
If you wish to speed up the rate at which potatoes are cooking in a pan of boiling water, would it be better to turn up the gas flame or use a pressure cooker?
-
C. 1) If the interest rates are .07 and .05 in Argentina and Canada respectively, what will happen to the peso versus the Canadian dollar, if presently they exchange as 10 pesos for one dollar? 2)...
-
Project ABC Initial End-of-Year Investment Cash Flows for years 1-3, respectively $36,000 $20,000 30,000 20,000 WACC = 13% What is the NPV? (Please round to the nearest dollar and do not enter the...
-
Vanscoy Bank pays 6.4% simple interest on its investment accounts. If Vade Bank pays interest on its accounts compounded annually, what rate should the bank set if it wants to match Vanscoy Bank over...

Study smarter with the SolutionInn App


