Determine the number of atoms per unit cell for each metal. (a) Polonium (c) Nickel (b) Tungsten
Question:
Determine the number of atoms per unit cell for each metal.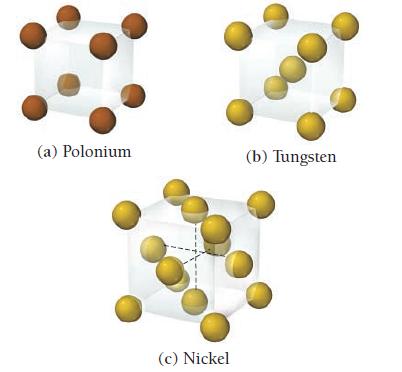
Transcribed Image Text:
(a) Polonium (c) Nickel (b) Tungsten
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a ...View the full answer

Answered By

Geoffrey Isaboke
I am an industrious tutor with a 5-yr experience in professional academic writing. I have passion for History and Music and I have good knowledge in Economics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Metallic lithium has a bee crystal structure. Each unit cell is a cube of side length a = 0.35nm. (a) For a bee lattice, what is the number of atoms per unit volume? Give your answer in terms of a....
-
Calculate the number of cells per unit volume in GaAs (a -5.65 A). Si has a 4% larger lattice constant. What is the unit cell density for Si? What is the number of atoms per unit volume in each case?
-
Zinc has the hexagonal closepacked crystal structure. The lattice parameters for zinc are a = 0.26648 nm and c = 0.49470 nm, and the atomic radius is 0.1332 nm. Note that zinc does not have the ideal...
-
11. What is the Specific Gravity of Zinc ? 12. The barometric pressure for the day is 14.7 psi. The hangar shop air tank gage reads 120 psi. What is the absolute pressure inside the shop air tank? =...
-
Harris, owner of certain land known as Red Bank, mailed a letter to Byron, a real estate broker in City X, stating, I have been thinking of selling Red Bank. I have never met you, but a friend has...
-
In CVP analysis gross margin is a less-useful concept than contribution margin . Do you agree? Explain briefly.
-
In 2016, a father of two minor children in Cuyahoga county, Ohio, filed a claim to determine custody of the children. At the pretrial, the father informed the magistrate he wanted to be named a legal...
-
On June 1 of the current year, Chad Wilson established a business to manage rental property. He completed the following transactions during June: a. Opened a business bank account with a deposit of...
-
In Milestone Two, you will review the Final Project Client Information document to identify all beneficiaries and discuss potential tax ramifications of receiving inherited property. In addition, you...
-
An X-ray beam of unknown wavelength is diffracted from a NaCl surface. If the interplanar distance in the crystal is 286 pm, and the angle of maximum reflection is found to be 7.23, what is the...
-
An X-ray beam with = 154 pm incident on the surface of a crystal produced a maximum reflection at an angle of = 28.3. Assuming n = 1, calculate the separation between layers of atoms in the crystal.
-
You've collected the following information about Fox, Inc.: Sales = $165,000 Net income = $19,000 Dividends = $4,500 Total debt = $49,000 Total equity = $72,000 What is the sustainable growth rate...
-
What happens if the semicolon after the SET statement in the following DATA st is missing?
-
What happens if a common area assessment is not voluntarily paid by a unit owner?
-
The seven classes of assets, which class related to accounts receivable, mortgages, credit card receivable which arise in the ordinary course of business?
-
Identify the basic provisions of the Sarbanes-Oxley Act that specifically deal with ethics and Independence and research how this Act has affected auditors since it was established in 2002. Be sure...
-
U Stores Ltd. had the following account balances at December 31, 2021: Deferred revenue $8,96 What is total net sales for this company?0 Travel and entertainment expense 11,340 Cash received from...
-
Assume that Amazon.com sells the MacBook Pro, a computer brand produced by Apple. Amazon arranges its operations such that customers receive products from Apple Stores rather than Amazon. Customers...
-
All of the following assets can be depreciated, except: (a) A bulldozer (b) A copper mine (c) A surgical robot (d) A conveyor belt
-
Myoglobin is a protein that participates in oxygen transport. For myoglobin in water at 20C, s = 2.04 10 13 s, D = 1.13 10 10 m 2 s 1 , and V = 0.740 cm g 1 . The density of water is 0.998 g cm 3...
-
The molecular weight of bovine serum albumin (BSA) is 66,500 g mol 1 and has a specific volume of 0.717 cm 3 g 1 . Velocity centrifugation demonstrates that s = 4.31 10 13 s for this protein....
-
You are interested in purifying a sample containing the protein alcohol dehydrogenase obtained from horse liver; however, the sample also contains a second protein, catalase. These two proteins have...
-
How do emergent properties such as resilience and stability manifest within ecological systems, and what are the underlying mechanisms that govern their dynamics in the face of environmental...
-
Provide one application of your choice, and argue that we would be better o, with a multithread architecture other than a multiprocess architecture when we design the application.?
-
What are the challenges and opportunities associated with integrating traditional ecological knowledge and indigenous perspectives into contemporary conservation efforts and natural resource...

Study smarter with the SolutionInn App


