Determine the products of each reaction. a. CH3 CH, C=CH, + H CH3 b. CH3 CH-CHCH-OH
Question:
Determine the products of each reaction.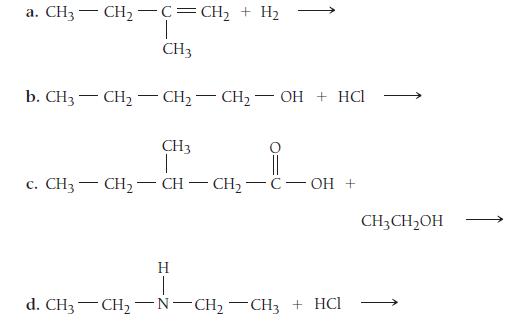
Transcribed Image Text:
a. CH3 — CH, C=CH, + Hạ CH3 b. CH3 CH₂-CH₂CH₂-OH + HCl CH3 1 c. CH3— CH,—CH–CH, C-OH + H 1 d. CH3 -CH₂-N-CH₂-CH3 + HCI CH3 CH₂OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
CH3 CH3 a H3C CH CH2 H H3C CH CH3 H 2methylbut1ene isopentane ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
It is generally claimed that state trading, or centrally controlled trading will tend to reach a lower economic welfare than would be reached by allowing market forces to determine trade flow...
-
Skillings Industrial Chemicals, Inc., operates a refinery in southwestern Ohio near the Ohio River. The companys primary product is manufactured from a chemical process that requires the use of two...
-
Brian is completing an insurance needs analysis for Becky and Stan Blombgren. Stan is a lawyer and a junior associate at a midsize law firm. Becky is a stay-at-home mom to their young daughter Josie....
-
For data Tables in an Oracle Server database, a blank field must be a. Indicated by NULL Avoided b. Kept as a blank c. Indicated either by NULL or a blank d. Indicated by NULL
-
Bebida Inc. produces soft drinks. Mixing is the first department and its output is measured in gallons. Bebida uses the FIFO method. All manufacturing costs are added uniformly. For August, the...
-
Explain how and why the process of budgeting used by multinational enterprises varies depending on the enterprise's home country.
-
In 1970, Rose Mary Knick purchased 90 acres of land in Scott Township, Lackawanna County, Pennsylvania. In 2008, another resident of Scott Township discovered documents that suggested that one of...
-
The Harriet Hotel in downtown Boston has 100 rooms that rent for $150 per night. It costs the hotel $30 per room in variable costs (cleaning, bathroom items, etc.) each night a room is occupied. For...
-
Step 3: Using the Price and Total Quantity Demanded data from the table above, graph the demand curve on the axes provided (be sure to provide numbers on the axes for price and quantity demanded)....
-
Draw the structure that corresponds to each name and indicate which structures can exist as stereoisomers. a. 3-methyl-1-pentene b. 3,5-dimethyl-2-hexene c. 3-propyl-2-hexene
-
How many kilograms of CO 2 does the complete combustion of 3.8 kg of n-octane produce?
-
How could we use the margin of safety to calculate the percent change in profit given a percent change in sales?
-
What is a risk management framework and why it should be linked to other business processes? As a Consultant illustrates how poor stakeholder's engagement on a Cervical & Prostate Screening Project...
-
Search the Internet and find three (3) accounting software applications that would be suitable for small-to-medium-sized businesses. Rachel Hayes is the owner of a digital marketing firm, has 12...
-
A VC invests 50,000 into a company at 200,000 pre-money valuation. What is the post-money valuation of the company?
-
please answe the question. Suppose the value 4 is removed from the following BST: Which of the trees below is the resulting BST?
-
When helping define the acquisition's requirements, why is it important to avoid short schedules and restrictive requirements? These practices make it more difficult to evaluate offerors These...
-
What two risks are present when acceptance sampling is used?
-
If a and b are positive numbers, find the maximum value of f ( x ) = x a (9 x ) b on the interval 0 x 9.
-
The magnetic field inside a 23-cm-diameter solenoid is increasing at 2.4 T/s. How many turns should a coil wrapped around the outside of the solenoid have so that the emf induced in the coil is 15 V?
-
Find the self-inductance of a 1500-turn solenoid 55 cm long and 4.0 cm in diameter.
-
The current in an inductor is changing at 110 A/s and the inductor emf is 45 V. Whats the self-inductance?
-
Cooper and Brandy are married and file a joint income tax return with two separate Schedule Cs. Cooper is an independent security specialist who spent $635 on uniforms during the year. His laundry...
-
Crystal Water, a community committed to raise awareness towards water pollution in big cities, permission approved to install CCC an innovation water cleaning device. Administrative costs to obtain...
-
Business Cs net profit is $12,923, depreciation is $24,286, and its working investment (the total of accounts receivable and inventory, less accounts payable and accrued expenses) increased $9,782....

Study smarter with the SolutionInn App


