Estimate the crystal field splitting energy (in kJ/mol) for a complex ion that is red in solution.
Question:
Estimate the crystal field splitting energy (in kJ/mol) for a complex ion that is red in solution.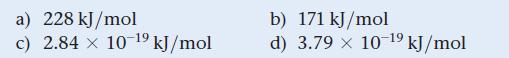
Transcribed Image Text:
a) 228 kJ/mol c) 2.84 x 10-¹9 kJ/mol b) 171 kJ/mol d) 3.79 x 10-¹⁹ kJ/mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
a ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1)How many geometric isomers are there for each species? Part A [Fe(CO)4Cl2] Part B [Pt(NH3)2Cl2Br2]+ 2) Which of the following complex ions absorbs light of the longestwavelength? Which of the...
-
The complex ion [Cu(NH 3 ) 6 ] 2 + is blue in aqueous solution. Estimate the crystal field splitting energy (in kJ/mol) for this ion.
-
Choose a DOW 30 company or you may already have it and predict their return (LN (price today/price yesterday) for today and compare it with the actual return on the stock price. Elaborate and perform...
-
Holly funded the Holly Marx Trust in January 2020. The entire trust income is payable to her adult son Jack for 20 years. At the end of the twentieth year, the trust assets are to pass to Hollys...
-
The reports that follow are from a grocery store. Which report would be used for financial purposes, and which would be used for activities-based decision making?Why? s 1,000 Scan grocery purchases...
-
Requirements Please read the attached case and respond to the questions below. Please keep your answers brief, to the point, and show all your calculations. a. Ignore any current plans. Using last...
-
In February 2014, defendant Ibrahim M. Shihadeh, d/b/a Creative Designs Kitchen and Baths, agreed to purchase 25% of his anticipated natural gas needs at a fixed price for the 201415 and 201516...
-
Julia Dumars is a licensed CPA. During the first month of operations of her business, Julia Dumars, Inc., the following events and transactions occurred. May 1 Stockholders invested $20,000 cash in...
-
Display Oracle database employee last_name and salary for employee_ids between 100and 102. Include a third column that divides each salary by 1.55 and rounds the result two decimal places.
-
Explain the differences between each pair of isomer types. a. Structural isomer and stereoisomer b. Linkage isomer and coordination isomer c. Geometric isomer and optical isomer
-
Using the Lewis acidbase definition, how would you categorize a ligand? How would you categorize a transition metal ion?
-
In many transition economies it is still the case that the price of electricity provided by the state utility is well below the marginal cost of production. Describe what is wrong in such a...
-
Criticality and sensitivity are determined in which step of the RMF?
-
Use the work of Cooper (2015), briefly explain why "prudence" ("conservatism") was restored within both the IASB's Exposure Drafts for a revised Conceptual Framework for Financial Reporting and the...
-
A woman, holding a dumbbell in each of her outstretched arms, stands at the center of a platform that is rotating at 1 rad/s. With her arms outstretched, the total moment of inertia the rotating...
-
What would be the anticipated outcome if the lowermost penny within a stack of pennies were to be displaced?
-
suppose AOL's Deffered Subscriber Acquisition Costs actually do generate benefits for AOL in 1998. Will AOL's net income for 1998 be understated, overstated or properly stated? Justify your opinion.
-
PMIS and cost accounting An Earned Value system can be a very effective way to monitor projects. However, Earned Value Analysis requires a Project Cost Accounting System (PCAS). (a) What are some of...
-
Ask students to outline the reasons why the various elements of culture (social structures and control systems, language and aesthetics, religion and other belief systems, educational systems, etc.)...
-
Two small particles have charges Q 1 = +3.0 C and Q 2 = -5.0 C. If the magnitude of the electric force between the particles is 120 N, what is the distance between the particles?
-
The particles in Problem 19 are conducting and are brought together so that they touch. Charge then moves between the two particles so as to make the excess charge on the two particles equal. If the...
-
Three charges with q = +7.5 ?C are located as shown in Figure P17.21, with L = 25 cm.? (a) What are the magnitude and direction of the total electric force on the charge at the bottom?? (b) What are...
-
Problem 12 A small and narrow conducting loop of area A and resistance R is positioned as shown relative to a long wire carrying current I, which is increasing with time. The distance from the wire...
-
#1. [4 points] To find the value of gravitational acceleration (g), a student uses a simple pendulum for which the time period (T) is given by T = 2 where I is the length of the pendulum. The student...
-
Interest income earned from investment Netflix Inc bonds and California municipal bonds. Is Angelo subject to the investment income tax rate of Netflix Inc bonds at 15% and California municipal bonds...

Study smarter with the SolutionInn App


