For each pair of substances, choose the one that you expect to have the higher standard molar
Question:
For each pair of substances, choose the one that you expect to have the higher standard molar entropy (S°) at 25 °C. Explain your choices.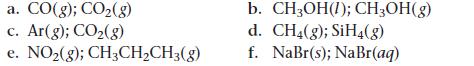
Transcribed Image Text:
a. CO(g); CO₂(g) c. Ar(g); CO₂(g) e. NO₂(g); CH3CH₂CH3(g) b. CH₂OH(1); CH3OH(g) d. CH₂(g); SiH4(g) f. NaBr(s); NaBr(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a CO 2 g greater molar mass and comple...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of substances, choose the one that you expect to have the higher standard molar entropy (S) at 25 C. Explain your choices. a. NaNO3(s); NaNO3(aq) c. Br(1); Br(g) e. PC13(g); PC15(g) b....
-
For each pair of substances listed here, choose the one having the larger standard entropy value at 25C. The same molar amount is used in the comparison. Explain the basis for your choice. (a) Li(s)...
-
For each pair of firms, choose the one that you think would be more sensitive to the business cycle. a. General Autos or General Pharmaceuticals. b. Friendly Airlines or Happy Cinemas.
-
Under what circumstance is it most appropriate to travel in reverse with a load? A. When you are carrying a non-standard load B. When a tall load obstructs your forward vision C. When the load is...
-
What are the objectives of economically developing countries? How do these objectives relate to marketing? Comment.
-
Pho Saigon Noodles and Catering accumulates the following adjustment data at December 31. 1. Services provided but unbilled total $750. 2. Food supplies of $300 have been used. 3. Utility expenses of...
-
Journalize the following adjusting entries at December 31: 1. Services provided but not recorded, $1,500. 2. Salaries earned by employees but not recorded, $2,300. 3. Accrued interest on a note...
-
On January 1, 2012, Valuation Allowance for Trading Investments had a zero balance. On December 31, 2012, the cost of the trading securities portfolio was $ 105,800 and the fair value was $101,600....
-
When It Comes to Eating Away at Democracy, Trump Is a Winner Links to an external site. . Thomas B. Edsall. The New York Times. 8/24/22 (segments of article) 1) After reading the article, how would...
-
What is the molar entropy of a pure crystal at 0 K? What is the significance of the answer to this question?
-
How does the molar entropy of a substance change with increasing temperature?
-
Let X1,..., Xn be a random sample from a n(, 2) population. Consider testing H0: 0 versus H1 : > 0. Let m denote the sample mean of the first m observations, X1,... ,Xm, for m = l,...,n. If 0 +...
-
.Harper Industries started its business in 2020, and its fiscal year ends on December 31. The following table provides information regarding Harper Industries" "Accounts Receivables": Accounts...
-
Define competitive advantage and discuss the concept from the perspective of global business. Often businesses use benchmarking to track performance as a way to assist improvement efforts. What are...
-
Month Total Units Produced Total Cost January 7,000 $21,200 February 8,300 $24,300 March 9,800 $26,810 April 7,900 $22,300 Use the high-low point method to determine total costs if 15,700 units are...
-
Pharoah Ltd. exchanged 460 common shares of Grants Corp. for new equipment from Milliken Sales. Pharoah was holding these shares as an investment and the shares were originally purchased for...
-
1. If 5 C of charge are moved through a +5V potential difference, determine the change in potential energy 2. A negatively charged particle (q = -2 C) moves through a 2000 V. Will there be a gain or...
-
Winter Companys balance sheet at December 31, 2016, is presented below. During January 2017, the following transactions occurred. Winter uses the perpetual inventory method. Jan. 1 Winter accepted a...
-
You are maintaining a subsidiary ledger account for Police-Training Expenditures for 2013. The following columns are used: Inventory purchases are initially recorded as expenditures. Record the...
-
A car starts from rest and then accelerates uniformly to a linear speed of 15 m/s in 40 s. If the tires have a radius of 25 cm, what are the magnitudes of (a) The average linear acceleration of a...
-
A car is traveling with a speed of 20 m/s. If the tires have an angular speed of 62 rad/s, what is the radius of the tires?
-
Consider a tennis ball that is hit by a player at the baseline with a horizontal velocity of 45 m/s (about 100 mi/h). The ball travels as a projectile to the players opponent on the opposite...
-
How do synthetic biology and metabolic engineering contribute to the development of novel bioprocessing systems for industrial biotechnology, and what are the implications for production efficiency...
-
What are the key factors in scaling up bioprocessing operations from laboratory to industrial scale, especially regarding bioreactor design, mass transfer, and process control ?
-
Respond to the following with why/why not you agree or disagree with the below statement. Even when a case seems cut and dry, every case deserves a trial. In our country, we are "innocent until...

Study smarter with the SolutionInn App


