How many copper atoms are in a copper penny with a mass of 3.10 g? (Assume that
Question:
How many copper atoms are in a copper penny with a mass of 3.10 g? (Assume that the penny is composed of pure copper.)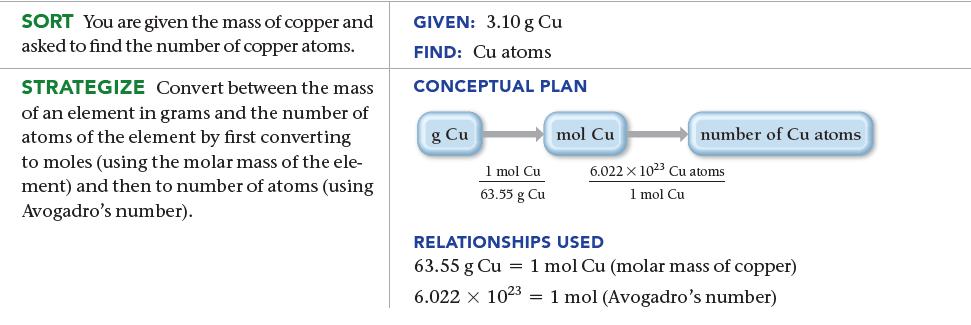
Transcribed Image Text:
SORT You are given the mass of copper and asked to find the number of copper atoms. STRATEGIZE Convert between the mass of an element in grams and the number of atoms of the element by first converting to moles (using the molar mass of the ele- ment) and then to number of atoms (using Avogadro's number). GIVEN: 3.10 g Cu FIND: Cu atoms CONCEPTUAL PLAN g Cu 1 mol Cu 63.55 g Cu mol Cu number of Cu atoms 6.022 x 1023 Cu atoms 1 mol Cu RELATIONSHIPS USED 63.55 g Cu = 1 mol Cu (molar mass of copper) 6.022 x 1023 1 mol (Avogadro's number)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
310 g x 1 mol ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Energy bands are considered continuous due to the large number of closely spaced energy levels. The range of energy levels in a crystal of copper is approximately 1 x 10 19 J. Assuming equal spacing...
-
Copper metal reacts with nitric acid. Assume that the reaction is 3Cu(s) + 8HNO3(aq) 3Cu(NO3)2(aq) + 2NO(g) + 4H2O(l) If 6.01 g Cu(NO3)2 is eventually obtained, how many grams of nitrogen monoxide,...
-
Metallic copper adopts an fcc structure with density 8960 kg m 3 . Draw the unit cell of copper and mark the shortest copper atom to copper atom distance. How many copper atoms are there in the unit...
-
IKEA was founded in 1943 by a 17-year-old Swede named Ingvar Kamprad. The company, which initially sold pens, Christmas cards, and seeds from a shed on Kamprad?s family farm, eventually grew into a...
-
What do the terms target cost and target price mean? Explain how they are developed.
-
Seven months ago, Naib Publishing Company published its first book (Book N). since then, Naib has added four more books to its product list (Books S, Q, X, and H). Management is considering proposals...
-
Is sustainable development a useful, practical perspective for business to adopt? If yes, how is it useful? Explain your view, giving reasons.
-
Michael McNamee is the proprietor of a property management company, Apartment Exchange, near the campus of Pensacola State College. The business has cash of $ 8,000 and furniture that cost $ 9,000...
-
Fact pattern Police witness a twenty-five year old man fleeing from a bank robbery scene. No one was injured during the robbery. The man is carrying a gun as he runs through the streets. They follow...
-
Using the What Do Operations Managers Do? box in the chapter, what key activities most directly relate to the case situation?
-
How and by whom was the electron discovered? What basic properties of the electron were reported with its discovery?
-
Determine the number of electrons in the Cr 3+ ion. a) 24 electrons b) 27 electrons c) 3 electrons d) 21 electrons
-
What are the various solution options regarding system development?
-
List and explain at least seven major types of OD interventions.
-
Which groups of managers are responsible for strategic, tactical, and operational plans?
-
How does the product life cycle affect the strategy employed by an SBU?
-
Discuss how leaders and managers differ.
-
Only one type of qualified opinion has qualifying language in both the scope paragraph and the opinion paragraph. What is the reason for this type of qualification?
-
On January 1, 2017, Black Jack Corporation purchases all of the preferred stock and 60% of the common stock of Zeppo Company for $56,000 and $111,000, respectively. Immediately prior to the...
-
An Atomic Energy Commission nuclear facility was established in Hanford, Washington, in 1943. Over the years, a significant amount of strontium 90 and cesium 137 leaked into the Columbia River. In a...
-
A compound with molecular formula C 4 H 6 O 2 has the following NMR spectrum. Determine the number of protons giving rise to each signal. Proton NMR 25 2.0 2.0 1.5 ppm 3.0 Integration Values 5.0 4.5...
-
The 1 H NMR spectrum of a compound with molecular formula C 7 H 15 C l exhibits two signals with relative integration 2 : 3. Propose a structure for this compound.
-
For each of the following compounds, determine the multiplicity of each signal in the expected 1 H NMR spectrum: (a) (b) (c) (d)
-
Issue 1 On 1 July 2021, we acquired an asset for $1,000,000, which it is depreciating using the straight-line method over 20 years (hence, $50,000 depreciation charged each year). We realised a...
-
What is the price of a European put with strike price $19 in 1 year? Calculate the price explicitly using a binomi dynamic hedging/replicating method, and verify that holds, together with your...
-
What are the differences between a finance lease and an operating lease? Why is the cost of replacement tires being ignored in the lease v buy analysis? Tori's textbook states that when making a...

Study smarter with the SolutionInn App


