How many types of hybrid orbitals do we use to describe each molecule? a. NO5 b. CH5NO
Question:
How many types of hybrid orbitals do we use to describe each molecule?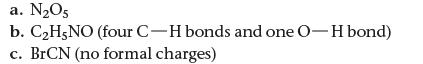
Transcribed Image Text:
a. N₂O5 b. C₂H5NO (four C-H bonds and one O-H bond) c. BrCN (no formal charges)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a ...View the full answer

Answered By

Rinki Devi
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and helped them achieve great subject knowledge.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Describe the various types of hybrid orbitals and how they are used to describe the bonding in alkanes, alkenes, and alkynes. How does hybridization explain that in allene, CH 2 =C=CH 2 , the two CH...
-
An atom in a molecule has two bonds to other atoms and one lone pair. What kind of hybrid orbitals do you expect for this atom? Describe how you arrived at your answer.
-
A school computer technician who was remotely accessing the hard drives of school-issued laptops for routine maintenance found sexually explicit images of a Grade 10 student on a high school...
-
Jeremiah Wedgewood, the CFO, is adamant that the company needs to move ahead with the new division. While Josey also thinks that creating a new division with a new product line is a good idea, she is...
-
Renfree Mines, Inc., owns the mining rights to a large tract of land in a mountainous area. The tract contains a mineral deposit that the company believes might be commercially attractive to mine and...
-
Calculating Liquidity Ratios SDJ, Inc., has net working capital of $l,570 current liabilities of $4,380, and inventory of $1,875. What is the current ratio? What is the quick ratio?
-
Tennessee law imposes durational-residency requirements on persons and companies wishing to operate retail liquor stores, requiring applicants for an initial license to have resided in the state for...
-
Data for Barry Computer Co. and its industry averages follow. a. Calculate the indicated ratios for Barry. b. Construct the DuPont equation for both Barry and the industry. c. Outline Barry's...
-
During the early 19th century, the Springfield (Massachu-setts) Armory provided a good example of: an increasing division of labor accounting techniques for wage payments and control of time and...
-
The most stable forms of the nonmetals in groups 4A, 5A, and 6A of the second period are molecules with multiple bonds. Beginning with the third period, the most stable forms of the nonmetals of...
-
The results of a molecular orbital calculation for NH 3 are shown here. Examine each of the orbitals and classify them as bonding, antibonding, or nonbonding. Assign the correct number of electrons...
-
Use the currency exchange rates in Table 3.2 to calculate the equivalent amounts of currencies. TABLE 3.2 Currency Cross Rates (noon, Toronto, August 13, 2009) Per C$ Per US$ Per Per Y Per Per AS...
-
During the Covid19 lockdown, yeasts popularity increased as millennials and home chefs were willing and able to spend more time cooking. Did yeasts increased popularity increase demand or the...
-
Why does economic growth destroy some jobs and create new jobs?
-
On the global PPF of healthcare supplies and other goods and services, how did the production point change as the Covid19 pandemic spread across the globe?
-
The table describes the preferences in Yucatan. a. What is the marginal benefit of sunscreen and how is it measured? b. Using the table in Problem 11 , what does Yucatan produce to achieve allocative...
-
How does economic growth change the patterns of production?
-
1. When you consider Milton Friedmans position on corporate responsibility in Chapter 4, is it possible to defend DPSs demand for lower hourly wages? 2. IS DPS considering the interests of all...
-
Use a calculator to evaluate the expression. Round your result to the nearest thousandth. V (32 + #)
-
The 2014-T6 aluminum rod has a diameter of 0.5 in. and is lightly attached to the rigid supports at A and B when T 1 = 70°F. Determine the force P that must be applied to the collar so that, when...
-
The 2014-T6 aluminum rod has a diameter of 0.5 in. and is lightly attached to the rigid supports at A and B when T 1 = 70°F. If the temperature becomes T 2 = 10°F, and an axial force of P =...
-
The force P is applied to the bar, which is made from an elastic perfectly plastic material. Construct a graph to show how the force in each section AB and BC (vertical axis) varies as P (horizontal...
-
What impact do healthcare policymakers have on the other stakeholders (patients, providers, and payors) from access, cost, and quality of care perspectives? Include at least one example in your...
-
Consider the following streaming version of linear regression algorithm. Algorithm StreamingSimpleLinearRegresssionModel 2 Double sumX = 0.0, sumY = 0.0, sumXY = 0.0, sumX = 0.0, a = 0.0, b = 0.0...
-
You have the following data on Stock A. Find the standard deviation. Economy Probability Return on A Bust 35% -10% Boom 65% 20%

Study smarter with the SolutionInn App


