Identify the correct balanced equation for the combustion of propane (C 3 H 8 ). a) C3H8(g)
Question:
Identify the correct balanced equation for the combustion of propane (C3H8).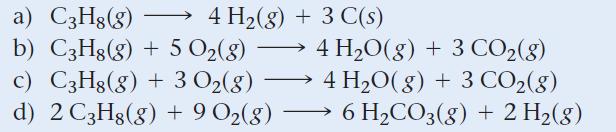
Transcribed Image Text:
a) C3H8(g) b) C3H8(g) + 5 0₂(8) c) C3H8(g) + 3 O₂(g) d) 2 C3H8(g) + 9 0₂(8) → 4 H₂(g) + 3 C(s) — - 4 H₂O(g) + 3 CO₂(g) → 4 H₂O(g) + 3 CO₂(g) 6 H₂CO3(g) + 2 H₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
b C3H...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
Propane C3H8 is burned with air, and the combustion products consist of CO2, CO, H2O, O2, N2, OH, H2, and NO. The number of equilibrium constant relations needed to determine the equilibrium...
-
A gasoline engine is converted to run on propane as shown in Fig. P13.144. Assume the propane enters the engine at 25C, at the rate of 40 kg/h. Only 90% theoretical air enters at 25C, so 90% of the C...
-
One item is omitted in each of the following summaries of balance sheet and income statement data for the following four different corporations: Determine the missing amounts, identifying them...
-
Zevon, Inc., has 9 percent coupon bonds on the market that have 8 years left to maturity. The bonds make annual payments. If the YTM on these bonds is 7 percent, what is the current bond price?
-
The W200 \(\times 46\) wide-flange A992-steel column can be considered pinned at its top and fixed at its base. Also, the column is braced at its mid-height against weak axis buckling. Determine the...
-
A process in which pressure remains constant is called (a) Isochoric process (b) Isobaric process (c) Isothermal process (d) Adiabatic process.
-
A craftsman named William Barnes builds two kinds of birdhouses, one for wrens and a second for bluebirds. Each wren birdhouse takes 4 hours of labor and 4 units of lumber. Each bluebird house...
-
If m = 3.8 kg resting on 20 o slope and string above the slope pulls parallel to the surface. What is the friction coefficient between the block m with the surface of the slope so the system will...
-
Let's say that professor Sanford dies (this is not going to happen for another 20 years), and the estate of decedent- Sanford comes into existence. Also, assume that you are the adopted child of...
-
Write a balanced equation for the combustion of liquid methyl alcohol (CH 3 OH).
-
In a chemical reaction, what is the theoretical yield and the percent yield?
-
Rank the following correlation coefficients on strength of their relationship (list the weakest first): +.71 +.36 -.45 +.47 -.62
-
In what ways can the different forms of communication (intrapersonal, interpersonal, small group, public and mass) be enhanced with the aid of technology?
-
Factor out the greatest common factor in the given polynomial. -3 -1 5(z+14) 3+ (z+14) 1 holow
-
On January 1st year 3, Terry's Board of Directors issued the management team 25,600 stock options for Terry's $1 par common stock. Terry's stock price on that day was $3.60/share. The Board set the...
-
Given sin 30 tan 30 =an and cos 30 = 2 determine the following:
-
Give examples of how society delivering or transmitting messages during the pre-media time.
-
The ISO commercial crime coverage form can be used to insure specific crime exposures of most business firms. Assume that you are a risk management consultant. For each of the following losses,...
-
Pearson Education, a publisher of college textbooks, would like to know if students prefer traditional textbooks or digital textbooks. A random sample of students was asked their preference and the...
-
If additional Cl 2 (g) is added to the reaction system at constant total pressure and temperature, how will the partial pressures of H 2 (g) and HCl(g) change?
-
Show that [(A/T)/(1/T)] V / U Write an expression analogous to Equation (6.36) that would allow you to relate A at two temperatures.
-
If additional Cl 2 (g) is added to the reaction system at constant V and T, how will the degree of dissociation of HCl(g) change
-
Given that 24 3 sin == 25' 2 == 4 3 < a < 2; tan =,
-
A review of the ledger of Blossom Company at December 31, 2022, produces the following data pertaining to the preparation of annual adjusting entries. 1. Prepaid Insurance $10,680. The company has...
-
Write an original summary of the article on how families can contribute negatively to college students' success outcomes. Summaries should not include direct quotations, instead use your own words to...

Study smarter with the SolutionInn App


