Identify the polyatomic ion and its charge in each compound: KNO 2 , CaSO 4 , Mg(NO
Question:
Identify the polyatomic ion and its charge in each compound: KNO2, CaSO4, Mg(NO3)2.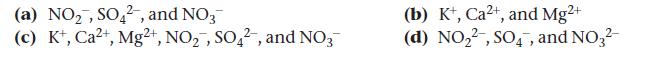
Transcribed Image Text:
(a) NO₂, SO42, and NO3 (c) K+, Ca2+, Mg2+, NO₂, SO42, and NO3 (b) K+, Ca²+, and Mg²+ (d) NO₂2, SO4, and NO3²-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Only this ...View the full answer

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When magnesium metal is burned in air (Figure 3.6), two products are produced. One is magnesium oxide, MgO. The other is the product of the reaction of Mg with molecular nitrogen, magnesium nitride....
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
Write a policy statement as the HR director stating whether or not office romantic relationships are allowed. If so, under what circumstances? What theoretical ethical perspective did you use to...
-
Melissa buys an iPod for $120 and gets consumer surplus of $80. a. What is her willingness to pay? b. If she had bought the iPod on sale for $90, what would her consumer surplus have been? c. If the...
-
What is the payback period for the following set of cash flows? Year Cash Flow 0 ....................... -$3,400 1 ....................... 1,200 2 ...................... 1,500 3...
-
An apparatus for measuring thermal conductivity employs an electrical heater sandwiched between two identical samples of diameter 30 mm and length 60 mm, which are pressed between plates maintained...
-
On January 2, 2018, The Register, a science and technology news site based in Britain, published an article revealing the existence of two major bugs leaving virtually all computers and smartphones...
-
Thom DeBusk, an architect, is considering buying, restoring, and reselling a home in the Draper-Preston historic district of Blacksburg, VA. The cost of the home is $240,000 and Thom believes it can...
-
Let G = (V,E) be a simple graph with |V| 2. The complement graph G of G is the simple graph whose vertex set is V and whose edge set consists of all the edges that have as endpoints nonadjacent...
-
On December 31, Y3, Aylmer Industries Inc. purchased 85% of the outstanding shares of Belmont Inc. The purchase price is indicated on the excel file. The entry has been recorded on the books of...
-
Name the compound Li 2 Cr 2 O 7 .
-
Explain how to write a formula for an ionic compound given the names of the metal and nonmetal (or polyatomic ion) in the compound.
-
The following questions relate to nonstatistical and monetary unit sampling. Choose the best response. a. A number of factors influence the sample size for a substantive test of details of an account...
-
List the environments in which shale might accumulate. What do all of these environments have in common? Some limestones are fine grained and black. How could you tell the difference between a...
-
Explain what Smith's book Desiring the Kingdom contributes to our understanding of communication today?
-
17. Suppose that during the TCP connection closing procedure, a machine that is in the TIME WAIT state crashes, reboots within MSL seconds, and immediately attempts to reestablish the connection...
-
Define the four key categories of preparation that leadership teams should consider for a successful strategic analysis. How do the 12 steps support a successful strategic planning process?
-
1. List the environments in which shale forms. What is the one thing that each has in common with the others? 2. What evidence would indicate that sandstones form on the Earth's surface and not...
-
Sidney Technologies Inc. assembles circuit boards by using a manually operated machine to insert electronic components. The original cost of the machine is $60,000, the accumulated depreciation is...
-
TRUE OR FALSE: 1. Banks with a significantly large share of fixed-interest rate home loans are less exposed to interest rate risks. 2. Although Australian banks are pretty big, they are not...
-
Predict the bond angles for all bonds in the following compounds: a) CH 3 CH 2 OH b) CH 2 O c) C 2 H 4 d) C 2 H 2 e) CH 3 OCH 3 f) CH 3 NH 2 g) C 3 H 8 h) CH 3 CN
-
Is the following statement correct? If not rewrite it so that it is correct. The superscript zero in H o f means that the reactions conditions are 298.15 K.
-
Why is it valid to add the enthalpies of any sequence of reactions to obtain the enthalpy of the reaction that is the sum of the individual reactions?
-
Give an Example NoSQL and describes is history and evolution in relation to databases?
-
3. A bag of mixed nuts contains almonds and hazelnuts. There are (6x+13) nuts in this particular bag, and (3x-7) of these are hazelnuts. Which expression represents the number of almonds in the bag?...
-
If V f(xz, y/z), prove that zV =xVx-yVy. =

Study smarter with the SolutionInn App


