List the products of each alcohol reaction. CH3 a. CH3-C- OH CH3 CH3 HSO4 NaCrO7 b. CH3
Question:
List the products of each alcohol reaction.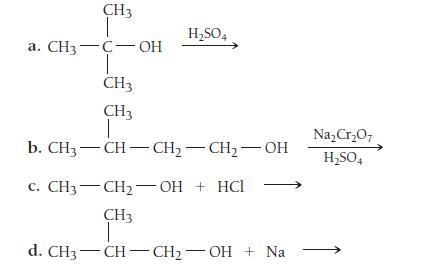
Transcribed Image Text:
CH3 a. CH3-C- OH CH3 CH3 H₂SO4 Na₂Cr₂O7 b. CH3 -CH-CH₂-CH₂-OH H₂SO4 C. CH3-CH₂-OH + HCl CH3 d. CH3 CH-CH₂-OH + Na
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The products of each alcohol reaction in the image are as follows a CH3COH H2SO4 CH3CO H...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
List the products of the reaction of 1-bromopropane with each of the following reagents. Write "no reaction" where appropriate. (Carefully evaluate the nucleophilic potential of each reagent.) (a)...
-
List the products of each alcohol reaction. a. CH3 CH-CH-OH b. CH3 -CH-CH-OH T CH3 c. CH3-CH-OH + Na CH3 + HBr HSO4 d. CH3-C-CH - CH-OH T CH3 NaCrO7 HSO4
-
Give an example reaction that would yield the following products as major organic products. For oxidation reactions, just write oxidation over the arrow and dont worry about the actual reagent. a....
-
A company has the following results for the three years to 31 October 2020: Assuming that all possible claims are made to relieve the trading loss against total profits, calculate the company's...
-
Describe the differences between process costing and job-order costing.
-
Explain how a manager might use the conceptual model of service quality to improve the quality of his/her own firm.
-
Management is considering three alternatives to satisfy an urgent need. Each of the alternatives will completely satisfy the need, so no combinations have to be considered. The first costs, operating...
-
1. Although the case with OFCCP is closed, we wonder if there are any less discriminatory explanations possible for why our women sales reps on average earned less than men. If so, what are they? 2....
-
Lab-created diamonds have become an alternative to traditionally mined diamonds. There are concerns, however, that these artificial diamonds may undermine diamond values and the reputation of natural...
-
Draw the structure for each alcohol. a. 2-butanol b. 2-methyl-1-propanol c. 3-ethyl-1-hexanol d. 2-methyl-3-pentanol
-
What are the products of each aromatic substitution reaction? a. b. H H H H H + Cl H FeCl3 CH3 I + CH3-C-Cl CH3 AIC13
-
On January 1, 20X5, Taft Company acquired all of the outstanding stock of Vikix, Inc., a Norwegian company, at a cost of $151,200. Vikixs net assets on the date of acquisition were 700,000 kroner...
-
ACCT 3140 Sp24 Case 1 Ratio Analysis Selected comparative statement data for Clare and Nick Company are presented here. All balance sheet data are as of December 31. Net sales Net income Total assets...
-
a. Match the terms (identified as a through g) with the definitions and phrases. For example, the term "a. Assets" matches with definition, "Economic resources that will be used by a business to...
-
Corrosion of metals in aerated aqueous environments involves the following cathodic reaction: (i) O2(aq) + 2H2O(l) +4e- 40H(aq) E = +0.401 V Calculate the equilibrium constant for the above cathodic...
-
9. What is(are) the output(s) of the following code fragment? string str1("Cat"), str2("Rat"), str3("Bat"), str4, str5, str6; str4 str1+str2; if(str1==str3) str5=str1; else str5=str2+str1;...
-
Clemson Software is considering a new project whose data are shown below. The required equipment has a 3-year tax life, after which it will be worthless. Under the new tax law, the equipment is...
-
What are the five areas of focus in the ISO 9000 principals?
-
APC16550D UART has a clock running at18.432 MHz and its baud rate is set to 2000.Determine the HEX contents of its DLM and DLL registers. Please can you explain step by step and in detail how you get...
-
An LC circuit with C = 18 mF undergoes oscillations with period 2.4 s. Find the inductance.
-
Your sister whos building the radio (Chapter 27 Problem 22) wants to use a variable capacitor with her toilet-paper-tube inductor to span the AM radio band (5501600 kHz). What capacitance range do...
-
A Driven RLC Circuits and Resonance 28. A series RLC circuit has R = 75 k, L = 20mH, and resonates at 4.0 kHz. (a) Whats the capacitance? (b) Find the circuits impedance at resonance and (c) at 3.0...
-
Bond X is a premium bond making semiannual payments. The bond has a coupon rate of 8.9 percent, a YTM of 6.9 percent, and has 14 years to maturity. Bond Y is a discount bond making semiannual...
-
The economy has recently entered a recession, and AutoEdge is near bankruptcy. What actions could the Federal Reserve take to stabilize the economy that would also help stabilize AutoEdge?
-
Question 4 (5 points) Listen According to the author, why are our friendships so important? Answer in two to three sentences and provide relevant paraphrased reference(s) from the reading in APA...

Study smarter with the SolutionInn App


